Key Takeaways
- AI-powered Electronic Lab Notebook software in 2026 is transforming labs into “active” research environments, enabling faster data interpretation and decision-making.
- Leading ELN platforms now unify ELN, LIMS, and data management systems, improving efficiency, compliance, and scalability across modern laboratories.
- Choosing the right ELN depends on factors like implementation speed, AI capabilities, regulatory compliance, and long-term ROI for research organizations.
The global scientific landscape in 2026 is undergoing a profound digital transformation, driven by the exponential growth of data, the rise of artificial intelligence, and the increasing complexity of research workflows. At the center of this transformation lies Electronic Lab Notebook (ELN) software, which has evolved far beyond its original purpose as a digital replacement for paper notebooks. Today, ELNs function as intelligent, cloud-native platforms that enable research organizations to capture, manage, analyze, and share data in real time, while ensuring strict regulatory compliance and operational efficiency.

As laboratories generate unprecedented volumes of structured and unstructured data across biotechnology, pharmaceuticals, chemistry, and academic research, the need for advanced digital infrastructure has become critical. Traditional data management approaches are no longer sufficient to support modern research demands. Instead, organizations are turning to next-generation ELN platforms that integrate Laboratory Information Management Systems (LIMS), Scientific Data Management Systems (SDMS), and AI-powered analytics into a unified ecosystem often described as a Lab Operating System. This convergence is redefining how scientific discoveries are made, validated, and scaled.
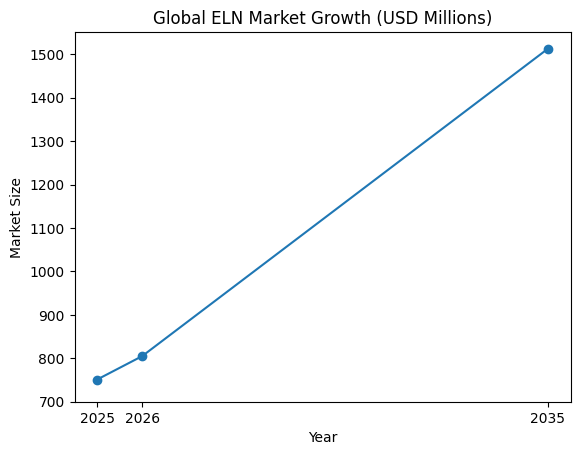
The Electronic Lab Notebook market in 2026 reflects this rapid evolution. With strong global adoption across both enterprise and emerging research organizations, ELN software is now considered a strategic investment rather than a simple operational tool. The market is expanding steadily, fueled by regulatory mandates, digital transformation initiatives, and the growing importance of data integrity in highly regulated industries. Pharmaceutical and biotechnology companies continue to lead adoption, but academic institutions, contract research organizations, and industrial laboratories are increasingly driving demand for flexible, scalable, and cost-effective solutions.
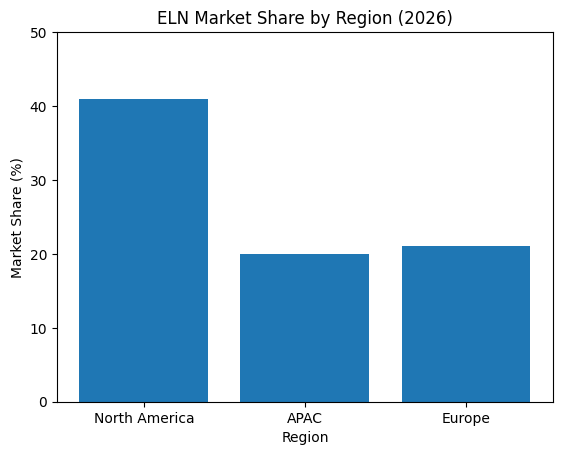
A key defining trend in 2026 is the emergence of AI-driven ELNs that actively assist scientists throughout the research lifecycle. These platforms are no longer passive repositories of experimental data. Instead, they act as intelligent research partners capable of interpreting results, automating documentation, generating insights, and even suggesting next steps in experimental design. This shift has significantly reduced the gap between data collection and decision-making, enabling faster innovation cycles and more efficient use of resources.
Another critical development is the widespread adoption of cloud-native architectures. Modern ELN platforms are designed to be accessible from anywhere, enabling seamless collaboration across global teams and research networks. This is particularly important in an era where scientific collaboration increasingly spans multiple institutions, geographies, and disciplines. Cloud-based deployment also reduces infrastructure costs and accelerates implementation timelines, allowing organizations to achieve faster return on investment and improved productivity.
At the same time, regulatory compliance remains a central consideration for ELN adoption. Standards such as 21 CFR Part 11 and EU Annex 11 require laboratories to maintain secure, traceable, and auditable records. Leading ELN platforms in 2026 are built with these requirements in mind, offering features such as audit trails, electronic signatures, and data provenance tracking. These capabilities ensure that digital records meet the stringent requirements of regulatory authorities, making ELNs indispensable for organizations operating in regulated environments.
The competitive landscape of ELN software in 2026 is highly dynamic, with a wide range of platforms catering to different use cases and organizational needs. Some solutions focus on AI-first innovation and rapid deployment, making them ideal for startups and agile research teams. Others prioritize deep functionality, scalability, and compliance, making them suitable for large enterprises with complex workflows. There are also platforms designed specifically for niche applications, such as chemistry research or biologics development, highlighting the increasing specialization within the market.
This comprehensive guide to the top 10 Electronic Lab Notebook software in the world in 2026 aims to provide a detailed and structured overview of the leading platforms shaping the future of laboratory research. It examines their core features, strengths, limitations, pricing models, and ideal use cases, enabling organizations to make informed decisions based on their unique requirements.
By understanding the capabilities and positioning of each platform, research leaders, laboratory managers, and decision-makers can identify the solutions that best align with their operational goals and long-term strategies. In an increasingly data-driven and competitive scientific environment, selecting the right ELN software is not just a technological choice—it is a strategic move that can significantly influence research outcomes, innovation speed, and overall organizational success.
Before we venture further into this article, we would like to share who we are and what we do.
About 9cv9
9cv9 is a business tech startup based in Singapore and Asia, with a strong presence all over the world.
With over nine years of startup and business experience, and being highly involved in connecting with thousands of companies and startups, the 9cv9 team has listed some important learning points in this overview of the Top 10 Electronic Lab Notebook Software To Use in 2026.
If you like to get your company listed in our top B2B software reviews, check out our world-class 9cv9 Media and PR service and pricing plans here.
Top 10 Electronic Lab Notebook Software To Use in 2026
- Genemod
- Benchling
- SciNote
- Labguru
- SciCord
- IDBS E-WorkBook
- Revvity Signals Notebook
- Dotmatics ELN
- SciSure (eLabNext)
- LabWare ELN
1. Genemod
In the global landscape of Electronic Lab Notebook (ELN) software in 2026, Genemod is widely positioned as a front-running platform that exemplifies the next generation of AI-driven laboratory systems. Built specifically for modern biopharma, diagnostics, and research-intensive environments, the platform is designed with an “AI-first” architecture, where artificial intelligence is deeply embedded into every layer of the system rather than functioning as a supplementary feature.
This architectural approach enables Genemod to unify ELN and LIMS functionalities within a single cloud-native ecosystem, eliminating traditional inefficiencies caused by fragmented laboratory tools. As research organizations increasingly transition toward digital-first operations, platforms like Genemod are enabling faster experimentation cycles, improved data traceability, and enhanced collaboration across distributed teams.
Unlike legacy systems that often require extensive onboarding timelines and complex infrastructure setup, Genemod is engineered for rapid deployment, allowing laboratories to operationalize within days. This capability significantly enhances time-to-value and supports organizations aiming to accelerate innovation and reduce operational friction.
Core AI Capabilities and Functional Differentiation
Genemod’s competitive advantage is anchored in its integrated AI ecosystem, which transforms how laboratory data is captured, analyzed, and utilized. These capabilities are interconnected, forming a cohesive framework that enhances productivity, reproducibility, and decision-making accuracy.
| Feature | Genemod Capability Description | Strategic Value for Laboratories |
|---|---|---|
| AI Chatbot Assistant | Conversational interface for querying experimental data and records | Accelerates data access and improves research efficiency |
| Automated Analysis Engine | Real-time interpretation of datasets with trend and anomaly detection | Enables faster, data-driven scientific decision-making |
| Protocol Template Generator | Auto-generates standardized protocols based on historical experiments | Improves consistency, compliance, and reproducibility |
| Experiment Summarization | Converts raw data into structured, publication-ready reports | Reduces documentation time and enhances reporting quality |
User Demographics and Market Penetration
Genemod’s adoption spans a diverse user base, reflecting its scalability across organizations of varying sizes and operational complexities. The platform has gained significant traction among both emerging biotech startups and large enterprise research organizations.
| Organization Type | Percentage Share | Key Insight |
|---|---|---|
| Small Businesses | 61.2% | Strong adoption among agile, innovation-driven startups |
| Enterprise Organizations | 34.7% | Increasing use in large-scale, complex research environments |
| Other Segments | 4.1% | Includes academic and niche research institutions |
Industry distribution further highlights its strong alignment with high-growth, research-intensive sectors:
| Industry Sector | Percentage Share | Strategic Relevance |
|---|---|---|
| Biotechnology | 53.0% | Core market with high demand for scalable ELN solutions |
| Research | 40.8% | Broad adoption across scientific and academic institutions |
| Other Industries | 6.2% | Includes healthcare, diagnostics, and adjacent sectors |
Customer Satisfaction and Market Perception
Genemod has achieved strong recognition across software review platforms, reflecting consistent user satisfaction and high perceived value.
| Performance Metric | Value | Interpretation |
|---|---|---|
| Overall Rating | 4.8 / 5.0 | Indicates excellent user satisfaction |
| Total Reviews | 49 | Solid validation across verified users |
| Five-Star Review Share | 87% | Strong endorsement of usability and performance |
These metrics reinforce Genemod’s positioning as a reliable, user-centric ELN platform with a strong emphasis on usability and innovation.
Pricing Structure and Scalability Model
Genemod adopts a tiered pricing strategy that aligns with the evolving needs of research organizations, from early-stage startups to enterprise-scale operations.
| Plan Tier | Target Users | Core Value Proposition |
|---|---|---|
| Free | Individual researchers and startups | Entry-level access to essential ELN functionalities |
| Team | Small research teams | Enhanced collaboration and workflow management tools |
| Business | Growing organizations | Advanced analytics, full AI suite, and operational insights |
| Enterprise | Large-scale organizations | Custom integrations, security controls, and dedicated support |
The Business plan is the most widely adopted tier, offering a comprehensive balance between advanced functionality and operational scalability.
Strategic Positioning Within the ELN Market in 2026
Within the broader ecosystem of top Electronic Lab Notebook software globally, Genemod represents a shift toward intelligent, automated, and user-centric laboratory platforms. Its AI-native foundation differentiates it from traditional ELN systems that rely heavily on manual workflows and disconnected data processes.
| Competitive Dimension | Traditional ELN Systems | Genemod AI-First Platform |
|---|---|---|
| Architecture | Add-on AI capabilities | Fully integrated AI-native system |
| Deployment Time | Months | Days |
| Data Analysis | Manual or semi-automated | Real-time, AI-driven interpretation |
| Workflow Integration | Fragmented ELN and LIMS systems | Unified, cloud-native environment |
| User Experience | Complex and technical | Simplified, intuitive, and accessible |
Key Strategic Insights for ELN Software Selection
Organizations evaluating ELN platforms in 2026 are increasingly prioritizing solutions that deliver measurable improvements in efficiency, scalability, and data intelligence.
| Decision Factor | Importance in 2026 ELN Market |
|---|---|
| AI-Native Architecture | Enables automation, predictive insights, and smarter workflows |
| Unified ELN + LIMS | Reduces system fragmentation and improves data consistency |
| Rapid Deployment | Minimizes downtime and accelerates research productivity |
| High User Satisfaction | Reflects usability and long-term adoption success |
| Flexible Pricing Models | Supports scalability across different growth stages |
Overall, Genemod stands out as a defining platform within the Top 10 Electronic Lab Notebook Software in the world in 2026, demonstrating how AI-first innovation is reshaping laboratory operations, accelerating scientific discovery, and setting new benchmarks for digital research infrastructure.
2. Benchling
Within the global landscape of Electronic Lab Notebook (ELN) software in 2026, Benchling continues to hold a dominant position as a highly specialized platform tailored for biotechnology, molecular biology, and life sciences research environments. Widely recognized as a foundational tool for biologics research, Benchling has established itself as a preferred solution among biotech startups, mid-sized research organizations, and enterprise-level pharmaceutical companies.
Its core strength lies in its deeply integrated molecular biology toolkit, which extends beyond traditional ELN capabilities. By combining experimental documentation with advanced sequence design, CRISPR guide development, and molecular cloning functionalities, Benchling enables researchers to operate within a unified digital environment. This seamless integration supports a more efficient transition from experimental planning to execution, significantly reducing workflow fragmentation and enhancing productivity across research pipelines.
Integrated Molecular Biology Capabilities and Workflow Efficiency
Benchling’s platform is designed to support complex biological research workflows, particularly in genomics, synthetic biology, and cell line development. Its ability to unify experimental design and execution within a single interface provides a substantial operational advantage.
| Core Capability | Functional Description | Strategic Benefit for Research Teams |
|---|---|---|
| Sequence Design Tools | Enables DNA, RNA, and protein sequence modeling and editing | Streamlines genetic engineering and molecular design |
| CRISPR Guide Design | Supports design and validation of CRISPR-based experiments | Accelerates gene editing workflows |
| Molecular Cloning Workflows | Integrated cloning simulation and construct design | Reduces experimental errors and improves reproducibility |
| Unified ELN Interface | Combines notebook documentation with experimental tools | Enhances workflow continuity and collaboration |
This integrated ecosystem positions Benchling as a powerful end-to-end platform for biologics research, particularly in environments where precision and traceability are critical.
Structured Data Architecture and Enterprise Scalability
A defining feature of Benchling is its structured data framework, which allows organizations to create customizable schemas for biological entities such as plasmids, cell lines, and reagents. This capability is particularly valuable for enterprise-scale research operations that require standardized data models and high levels of data integrity.
| Data Capability | Description | Enterprise Impact |
|---|---|---|
| Custom Entity Schemas | Configurable data models for biological materials | Enables standardized data management across teams |
| Centralized Data Repository | Unified storage of experimental and biological data | Improves accessibility and collaboration |
| Data Traceability | Tracks changes and experimental lineage | Supports regulatory compliance and audit readiness |
However, this structured approach introduces a notable challenge: data portability. Many users report difficulties in migrating complex datasets out of Benchling, leading to concerns around vendor lock-in, particularly for organizations seeking long-term flexibility across systems.
Pricing Model and Accessibility Considerations
Benchling’s pricing structure reflects its positioning as a premium, enterprise-grade platform. While it delivers substantial value for large organizations, the cost can be prohibitive for smaller laboratories and early-stage startups.
| Pricing Metric | Benchling Performance | Market Implication |
|---|---|---|
| Annual Pricing | $5,000 – $7,000 per user | High cost barrier for smaller organizations |
| Value Proposition | Comprehensive biologics toolkit | Justifies cost for advanced research environments |
This pricing model reinforces Benchling’s focus on high-value research environments but limits accessibility for cost-sensitive users.
User Demographics and Market Positioning
Benchling’s user base reflects strong adoption across innovation-driven organizations, particularly within the biotechnology sector.
| Market Segment | Percentage Share | Key Insight |
|---|---|---|
| Small Businesses | 63% | Strong adoption among biotech startups and early-stage firms |
| Mid-Market | 31% | Widely used by scaling research organizations |
| Enterprise | 6% | Select adoption among large pharmaceutical companies |
This distribution highlights Benchling’s role as a growth-stage platform that supports organizations transitioning from early research to scaled operations.
Performance Metrics and Reliability Standards
Benchling maintains strong performance benchmarks and reliability standards, which are critical for mission-critical research environments.
| Performance Metric | Benchling Performance | Strategic Importance |
|---|---|---|
| Satisfaction Score | 4.5 / 5.0 (61 reviews) | Indicates strong user approval and platform reliability |
| Service Level Agreement | 99.9% uptime | Ensures consistent system availability |
| Compliance Standards | 21 CFR Part 11; EU Annex 11 | Supports regulatory compliance in pharma environments |
| Security Certifications | ISO 27001:2022; SOC 2 Type 2 | Meets global enterprise security requirements |
These credentials reinforce Benchling’s suitability for regulated industries, particularly in pharmaceutical and clinical research settings.
Limitations and Functional Gaps
Despite its leadership in biologics, Benchling exhibits certain limitations that may impact its suitability for broader scientific applications.
| Limitation Area | Description | Impact on Users |
|---|---|---|
| Chemistry Capabilities | Limited support for chemistry-focused workflows | Less suitable for chemical research laboratories |
| Data Portability | Challenges in exporting structured data | Creates long-term dependency on the platform |
| Pricing Accessibility | High cost per user | Restricts adoption among smaller research teams |
For chemistry-intensive laboratories, alternative platforms such as Dotmatics or Signals Notebook may offer more specialized functionality.
Strategic Positioning in the Global ELN Market
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, Benchling is positioned as a biologics-focused powerhouse that excels in integrating experimental design with execution. Its strength lies in enabling data-driven, highly structured research workflows, particularly in genomics and synthetic biology.
| Competitive Dimension | Benchling Positioning | Market Advantage |
|---|---|---|
| Core Focus | Biologics and molecular biology | Deep specialization in life sciences research |
| Platform Integration | ELN + molecular design tools | End-to-end research workflow support |
| Target Market | Biotech startups and scaling organizations | Strong alignment with high-growth sectors |
| Enterprise Readiness | High | Meets compliance and security standards |
Key Insights for Decision-Makers
Organizations evaluating ELN platforms in 2026 can derive several strategic insights from Benchling’s positioning:
| Decision Factor | Strategic Consideration |
|---|---|
| Specialization vs Flexibility | Benchling excels in biologics but may not suit all disciplines |
| Data Architecture | Structured data enhances control but may limit portability |
| Cost vs Value | High investment justified for advanced research capabilities |
| Compliance Requirements | Strong regulatory support for pharmaceutical use cases |
Benchling remains one of the most influential platforms in the ELN ecosystem, particularly for biologics-driven research organizations. Its ability to integrate molecular design with experimental workflows positions it as a critical tool for advancing scientific innovation, even as organizations carefully weigh considerations around cost, flexibility, and long-term data strategy.
3. SciNote
In the global Electronic Lab Notebook (ELN) landscape in 2026, SciNote has established itself as a leading platform specifically tailored for academic institutions, government laboratories, and research organizations transitioning from traditional paper-based documentation systems. Known for its exceptional usability and accessibility, SciNote is widely recognized as one of the easiest ELN platforms to adopt, making it a preferred choice among universities, public research agencies, and regulatory bodies.
Its adoption by prominent organizations, including federal agencies such as the FDA and USDA, reflects its credibility and reliability in structured, compliance-driven environments. With a global user base exceeding 90,000 researchers across more than 100 countries, SciNote demonstrates strong international penetration and widespread trust within the scientific community.
Unlike highly complex enterprise ELN systems, SciNote focuses on delivering a streamlined, intuitive experience that simplifies laboratory digitization while maintaining essential functionality for data capture, project tracking, and inventory management.
Core Functional Capabilities and Workflow Optimization
SciNote offers an all-in-one laboratory management solution that integrates multiple research workflows into a cohesive and user-friendly platform. Its design prioritizes ease of navigation, minimal training requirements, and seamless adoption.
| Core Functionality | Description | Operational Benefit |
|---|---|---|
| Inventory Management | Tracks lab materials, reagents, and equipment | Improves resource visibility and reduces waste |
| Project Tracking | Organizes experiments and research workflows | Enhances project coordination and accountability |
| Data Capture | Digital recording of experimental data | Eliminates paper-based inefficiencies |
| Protocol Templates | Pre-built and customizable experiment templates | Standardizes processes and improves reproducibility |
This integrated approach enables laboratories to centralize their operations while maintaining simplicity and efficiency.
Operational Efficiency and Implementation Metrics
SciNote’s operational efficiency is one of its strongest differentiators, particularly for organizations seeking rapid adoption with minimal disruption.
| Operational Metric | SciNote Performance | Strategic Insight |
|---|---|---|
| Implementation Timeline | Approximately 2 months | Faster onboarding compared to legacy ELN systems |
| Customer Satisfaction | 99% | Indicates high usability and strong support experience |
| Primary Benefits | Ease of Use, Organization Efficiency | Reflects strong alignment with user needs |
The platform’s intuitive interface significantly reduces the learning curve, allowing research teams to quickly transition from manual workflows to fully digital systems.
User Satisfaction and Performance Ratings
SciNote consistently achieves strong ratings across multiple usability and functionality categories, reinforcing its reputation as a user-friendly ELN platform.
| Satisfaction Category | SciNote Rating (Average: 8.8) | Category Benchmark Comparison |
|---|---|---|
| Ease of Use | 8.8 | High usability across all user segments |
| Protocol Templates | 8.8 | Above category average (8.6) |
| Data Storage | 8.8 | Slightly below category average (9.1) |
| Data Access Authorization | 8.8 | Competitive with category average (8.9) |
These ratings demonstrate balanced performance across critical ELN functionalities, particularly in usability and workflow standardization.
Compliance Capabilities and Limitations
SciNote supports essential regulatory compliance requirements, making it suitable for many academic and government use cases. However, its capabilities are more foundational compared to advanced enterprise-grade platforms.
| Compliance Feature | SciNote Capability | Implication for Users |
|---|---|---|
| 21 CFR Part 11 | Basic compliance | Suitable for regulated environments with moderate needs |
| Good Laboratory Practice | Supported | Enables standardized research documentation |
| Advanced AI Analytics | Not available | Limited predictive and automated insights |
While SciNote provides sufficient compliance support for many institutions, it does not offer the advanced AI-driven analytics found in next-generation ELN platforms such as Genemod or Scispot.
Pricing Model and Accessibility
SciNote adopts a flexible pricing strategy designed to support a wide range of users, from individual researchers to institutional deployments.
| Pricing Tier | Target Users | Key Value Proposition |
|---|---|---|
| Free Version | Individual researchers and small labs | Entry-level access with core ELN functionalities |
| Premium Academia | Universities and academic labs | Enhanced collaboration and academic-focused features |
| Industry Plan | Commercial and private research labs | Expanded capabilities and enterprise-level support |
This tiered approach makes SciNote one of the most accessible ELN platforms in the market, particularly for cost-sensitive organizations.
Integration Ecosystem and Workflow Continuity
SciNote’s ability to integrate with widely used software tools enhances its practicality and ease of adoption within existing research environments.
| Integration Tool | Functionality | User Benefit |
|---|---|---|
| Microsoft 365 | Integration with Word and Excel | Maintains continuity with familiar productivity tools |
| Quartzy | Lab supply and inventory management integration | Streamlines procurement and inventory workflows |
These integrations allow researchers to maintain established workflows while gradually transitioning to a fully digital laboratory environment.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, SciNote is positioned as a highly accessible, user-centric platform that excels in ease of use and operational simplicity.
| Competitive Dimension | SciNote Positioning | Market Advantage |
|---|---|---|
| Core Focus | Academic and government research | Strong alignment with public sector needs |
| Ease of Use | Industry-leading | Minimal training required for adoption |
| Feature Complexity | Moderate | Balanced functionality without overwhelming users |
| AI Capabilities | Limited | Focus on usability rather than advanced analytics |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several important considerations from SciNote’s positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Ease of Adoption | Critical for institutions transitioning from paper-based systems |
| Cost Accessibility | Important for academic and publicly funded organizations |
| Compliance Requirements | Adequate for moderate regulatory environments |
| Advanced Analytics Needs | May require alternative platforms for AI-driven insights |
SciNote stands out as a highly practical and accessible ELN solution in 2026, particularly for academic institutions and government laboratories seeking to digitize their research workflows without the complexity of enterprise-grade systems. Its emphasis on usability, affordability, and integration makes it a strong contender within the global ELN market, especially for organizations prioritizing simplicity and operational efficiency over advanced AI-driven capabilities.
4. Labguru
In the global Electronic Lab Notebook (ELN) software landscape in 2026, Labguru has positioned itself as a comprehensive, all-in-one platform that effectively bridges the gap between ELN, Laboratory Information Management Systems (LIMS), and laboratory informatics. Designed to support end-to-end laboratory operations, Labguru enables research organizations to centralize experimental data, operational workflows, and resource management within a single cloud-hosted environment.
This unified approach makes Labguru particularly valuable for life sciences organizations that require tight integration between experimental documentation, inventory systems, and laboratory processes. By consolidating multiple systems into one platform, Labguru reduces operational silos and enhances data consistency across research teams.
Core Platform Capabilities and Functional Integration
Labguru is designed to manage the full spectrum of laboratory activities, offering a robust set of features that support both scientific research and operational management.
| Core Capability | Functional Description | Strategic Value for Laboratories |
|---|---|---|
| Electronic Lab Notebook | Centralized documentation of experiments and research data | Improves data traceability and collaboration |
| LIMS Integration | Workflow management and sample tracking | Enhances operational efficiency and standardization |
| Inventory Management | Tracks reagents, materials, and lab supplies | Reduces waste and ensures resource availability |
| Equipment Management | Monitors and manages laboratory instruments | Improves utilization and maintenance planning |
| SOP Management | Stores and standardizes procedures | Supports compliance and reproducibility |
This holistic functionality allows laboratories to operate within a single digital ecosystem, minimizing the need for multiple disconnected tools.
Adoption Trends and Market Penetration
Labguru has achieved strong adoption among small and mid-sized organizations, reflecting its suitability for growing research environments that require scalable and integrated solutions.
| Organization Segment | Percentage Share | Key Insight |
|---|---|---|
| Small Businesses | 75% | Strong adoption among agile and resource-conscious labs |
| Mid-Market | 19% | Growing usage among scaling research organizations |
| Enterprise | 6% | Limited but increasing adoption in larger institutions |
This distribution highlights Labguru’s appeal as a scalable platform for organizations transitioning from basic ELN systems to more integrated laboratory management solutions.
Performance Metrics and User Satisfaction
Labguru maintains strong user satisfaction ratings, particularly in areas related to data management and workflow integration.
| Performance Metric | Labguru Score | Category Average | Performance Insight |
|---|---|---|---|
| Data Storage | 8.8 | 9.1 | Slightly below average but still highly competitive |
| Protocol Templates | 8.6 | 8.6 | On par with industry standards |
| Data Access Authorization | 8.7 | 8.9 | Strong but marginally below benchmark |
| Ease of Use | 8.4 | 8.8 | Lower than average due to steeper learning curve |
Overall, Labguru holds a user rating of 4.6 out of 5, indicating strong overall satisfaction despite some usability challenges.
Advanced Scientific Capabilities and Data Insights
One of Labguru’s distinguishing strengths lies in its ability to provide deeper insights into laboratory data creation, particularly within chemistry and molecular biology domains.
| Advanced Feature | Description | Strategic Benefit |
|---|---|---|
| Chemistry Data Resources | Tools tailored for chemical experiment tracking and analysis | Enhances research precision in chemistry workflows |
| Molecular Biology Support | Integrated resources for biological experiment management | Supports complex life sciences research |
| Data Insight Generation | Analytical capabilities for understanding data creation patterns | Improves experimental planning and optimization |
These capabilities position Labguru as a versatile platform capable of supporting multidisciplinary research environments.
Limitations and Operational Constraints
Despite its comprehensive feature set, Labguru presents certain limitations that may impact its effectiveness in high-throughput or highly automated laboratory environments.
| Limitation Area | Description | Impact on Organizations |
|---|---|---|
| Learning Curve | More complex interface compared to simpler ELN platforms | Requires additional training and onboarding time |
| Instrument Integration | Lack of real-time integration with high-throughput instruments | Limits automation for data-intensive laboratories |
| Scalability for Automation | Challenges in handling large volumes of automated data | Potential bottleneck for advanced research facilities |
These constraints suggest that while Labguru is highly effective for integrated lab management, it may require supplementary tools in environments with heavy automation demands.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, Labguru is positioned as a comprehensive, integration-focused platform that excels in unifying laboratory operations.
| Competitive Dimension | Labguru Positioning | Market Advantage |
|---|---|---|
| Core Focus | Integrated ELN + LIMS + informatics | All-in-one laboratory management solution |
| Target Market | Small to mid-sized research organizations | Strong alignment with growing labs |
| Feature Depth | High | Comprehensive functionality across lab operations |
| Ease of Use | Moderate | More complex than entry-level ELN platforms |
| Automation Capability | Limited | Not optimized for high-throughput environments |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic insights from Labguru’s positioning:
| Decision Factor | Strategic Consideration |
|---|---|
| Integration Needs | Ideal for labs seeking unified ELN and LIMS functionality |
| Ease of Adoption | Requires more training compared to simpler platforms |
| Data Complexity | Well-suited for managing structured and diverse datasets |
| Automation Requirements | May require additional tools for high-throughput workflows |
Labguru stands out as a powerful, all-in-one ELN platform that delivers strong value through integration and comprehensive functionality. Its ability to centralize laboratory operations makes it a compelling choice for life sciences organizations seeking efficiency and scalability, even as users must consider its learning curve and limitations in highly automated environments.
5. SciCord
In the global Electronic Lab Notebook (ELN) software market in 2026, SciCord has emerged as a highly efficient and cost-effective solution specifically designed for regulated analytical and quality control (QC) laboratories. Positioned as a unified ELN and Laboratory Information Management System (LIMS), SciCord addresses the growing demand for platforms that can simultaneously ensure compliance, streamline workflows, and improve operational efficiency.
Its strongest differentiator lies in its ability to support laboratories transitioning from traditional tools such as paper-based records and spreadsheet systems. By offering a familiar, spreadsheet-like interface, SciCord allows researchers to continue using existing workflows while enhancing them with advanced digital capabilities such as audit trails, validated formulas, and compliance-ready documentation.
This approach significantly lowers the barrier to adoption, making SciCord an attractive option for regulated environments that require both precision and ease of use.
Core Platform Capabilities and Workflow Optimization
SciCord is engineered to deliver a seamless integration of ELN and LIMS functionalities, enabling laboratories to manage both experimental data and operational processes within a single system.
| Core Capability | Functional Description | Strategic Value for Regulated Labs |
|---|---|---|
| Unified ELN + LIMS | Combines experiment documentation with workflow and sample tracking | Eliminates system fragmentation and improves data consistency |
| Spreadsheet-Like Interface | Familiar grid-based environment for data entry and analysis | Reduces training time and accelerates user adoption |
| Audit Trails | Tracks all changes and user actions | Ensures compliance and audit readiness |
| Validated Formulas | Built-in calculation validation for regulated processes | Improves accuracy and reduces compliance risks |
These features collectively enable laboratories to digitize their operations without disrupting established workflows.
Pricing Model and Return on Investment
SciCord is widely regarded as one of the most cost-effective platforms in the ELN market, offering significant value compared to competing enterprise-grade solutions.
| Economic Metric | SciCord Performance | Strategic Implication |
|---|---|---|
| Monthly Pricing | Approximately $220 per user | Highly competitive pricing relative to enterprise solutions |
| Implementation Timeline | Measured in weeks | Rapid deployment reduces operational downtime |
| Documentation Efficiency | 3x improvement | Significant productivity gains across research workflows |
| Comparative Cost Advantage | 3x to 10x lower than competitors | Strong value proposition for budget-conscious organizations |
This pricing structure makes SciCord particularly attractive for laboratories seeking enterprise-level functionality without the associated high costs.
Compliance and Regulatory Readiness
SciCord is specifically designed to meet the stringent requirements of regulated industries, making it suitable for pharmaceutical, clinical, and quality control laboratories.
| Compliance Feature | SciCord Capability | Regulatory Impact |
|---|---|---|
| FDA Audit Performance | Audited twice with no findings | Demonstrates high compliance reliability |
| Audit Trail System | Fully integrated | Ensures traceability and accountability |
| Validated Data Handling | Built-in formula validation | Supports regulatory standards such as GxP |
These capabilities position SciCord as a trusted platform for organizations operating in highly regulated environments.
AI and Data Intelligence Capabilities
In addition to its compliance-focused features, SciCord incorporates native AI and machine learning capabilities that enhance data interpretation and reduce reliance on external tools.
| AI Capability | Description | Strategic Benefit |
|---|---|---|
| Native AI/ML Integration | Built-in tools for analyzing and interpreting laboratory data | Reduces need for third-party analytics platforms |
| Data Pattern Recognition | Identifies trends and anomalies within datasets | Improves decision-making and experimental outcomes |
| Workflow Optimization | Uses data insights to improve lab processes | Enhances efficiency and resource utilization |
These features enable laboratories to move toward more data-driven operations while maintaining compliance and control.
Pricing Flexibility and Scalability
SciCord adopts a transparent and scalable pricing model that includes all core functionalities by default, simplifying budgeting and procurement processes.
| Pricing Dimension | Description | Benefit for Organizations |
|---|---|---|
| Inclusive Feature Model | All functionalities included in base pricing | Eliminates hidden costs and simplifies decision-making |
| User-Based Scaling | Costs increase with number of users | Supports organizational growth |
| Compliance-Based Scaling | Pricing varies based on regulatory requirements | Aligns costs with operational complexity |
This model ensures that organizations can scale their usage without encountering unexpected costs or limitations.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, SciCord is positioned as a high-efficiency, compliance-driven platform that delivers exceptional value for regulated laboratories.
| Competitive Dimension | SciCord Positioning | Market Advantage |
|---|---|---|
| Core Focus | Regulated analytical and QC labs | Strong alignment with compliance-heavy environments |
| Ease of Adoption | High | Familiar interface reduces onboarding complexity |
| Cost Efficiency | Very high | Significantly lower cost than comparable solutions |
| AI Capabilities | Integrated | Enhances data analysis without external tools |
| Deployment Speed | Fast | Enables rapid digital transformation |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic considerations from SciCord’s positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Compliance Requirements | Critical for regulated industries |
| Cost vs Value | SciCord offers strong ROI compared to premium competitors |
| Ease of Transition | Ideal for labs migrating from spreadsheets or paper systems |
| Data Intelligence Needs | Built-in AI reduces reliance on external analytics tools |
SciCord stands out as a highly practical and value-driven ELN solution in 2026, particularly for regulated laboratories seeking to modernize their workflows without incurring excessive costs or operational complexity. Its combination of affordability, compliance readiness, and user-friendly design makes it a compelling choice within the global ELN market, especially for organizations prioritizing efficiency, scalability, and regulatory assurance.
6. IDBS E-WorkBook
In the global Electronic Lab Notebook (ELN) software ecosystem in 2026, IDBS E-WorkBook stands as one of the most established and trusted enterprise-grade platforms, particularly within large pharmaceutical companies and highly regulated research and development environments. Backed by Danaher, IDBS benefits from decades of scientific software expertise, with a legacy spanning over 35 years in serving compliance-driven industries.
The platform is widely recognized for its template-driven architecture, which enables organizations to standardize experimental workflows while ensuring strict adherence to regulatory requirements such as GxP compliance. This makes IDBS E-WorkBook a preferred choice for global pharmaceutical leaders that require absolute data integrity, auditability, and consistency across complex, multi-site research operations.
Core Platform Capabilities and Enterprise Functionality
IDBS E-WorkBook is designed to support large-scale, highly regulated R&D environments, offering a robust set of features focused on standardization, compliance, and data governance.
| Core Capability | Functional Description | Strategic Value for Enterprises |
|---|---|---|
| Template-Driven ELN | Standardized experiment templates across research workflows | Ensures consistency and regulatory compliance |
| Data Integrity Framework | Built-in validation and audit mechanisms | Supports GxP and regulatory audit readiness |
| Centralized Data Management | Unified repository for structured scientific data | Enhances collaboration and traceability |
| Workflow Standardization | Pre-configured processes for regulated environments | Reduces variability and improves operational efficiency |
These capabilities make IDBS particularly suitable for organizations operating in highly controlled environments where compliance and reproducibility are critical.
Market Adoption and Global Reach
IDBS E-WorkBook demonstrates strong adoption among leading pharmaceutical organizations, reinforcing its position as an enterprise-standard ELN solution.
| Adoption Metric | IDBS Performance | Strategic Insight |
|---|---|---|
| Global Pharma Reach | Used by 18 of the top 20 companies | Indicates dominance among industry leaders |
| Market Position | Most widely used platform in comparative study | Strong validation across enterprise environments |
| Customer Segment | Large pharmaceutical and enterprise R&D | Focused on high-complexity, regulated use cases |
This level of adoption underscores its credibility and reliability in mission-critical research environments.
Performance Metrics and Operational Impact
IDBS E-WorkBook delivers measurable improvements in research efficiency and data management, as demonstrated through real-world case studies.
| Impact Metric | Performance Outcome | Business Impact |
|---|---|---|
| Study Cycle Time Reduction | 30% reduction | Accelerates research timelines and product development |
| Data Input Time Reduction | 75% reduction | Significantly improves operational efficiency |
| Net Promoter Score (NPS) | Highest in category study | Reflects strong customer satisfaction and loyalty |
| Integration Capability | Over 300 connectors (REST API, SQL) | Enables seamless integration with enterprise systems |
These metrics highlight the platform’s ability to drive efficiency gains at scale while maintaining strict compliance standards.
Cloud Evolution and AI/ML Readiness
The introduction of the Polar platform in 2020 marked a significant evolution for IDBS, transitioning it toward a more modern, cloud-native architecture.
| Platform Enhancement | Description | Strategic Advantage |
|---|---|---|
| Polar Cloud Platform | Cloud-native infrastructure for scalability | Improves accessibility and reduces on-premise dependency |
| Open Data Model | Flexible data architecture for integration and analytics | Supports advanced data interoperability |
| AI/ML Readiness | Enables integration with machine learning tools | Prepares organizations for data-driven research innovation |
These advancements position IDBS as a forward-looking platform capable of supporting emerging trends in AI-driven scientific research.
System Complexity and Implementation Considerations
Despite its extensive capabilities, IDBS E-WorkBook is often characterized as a complex and resource-intensive system, particularly when compared to newer, more agile ELN platforms.
| Complexity Factor | Description | Impact on Organizations |
|---|---|---|
| Implementation Effort | Requires extensive configuration and validation | Longer deployment timelines |
| IT Resource Requirements | High dependency on technical expertise | Increased operational overhead |
| User Training | Steep learning curve for end users | Additional onboarding time and costs |
| System Flexibility | Less agile compared to modern platforms | Slower adaptation to evolving research needs |
Deployment typically involves service-heavy validation cycles, particularly in regulated environments where compliance requirements must be rigorously met.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, IDBS E-WorkBook is positioned as the enterprise gold standard for regulated R&D environments, offering unmatched reliability, compliance, and scalability.
| Competitive Dimension | IDBS Positioning | Market Advantage |
|---|---|---|
| Core Focus | Enterprise pharmaceutical R&D | Deep alignment with regulated industries |
| Compliance Strength | Very high | Industry-leading support for GxP environments |
| Integration Capability | Extensive | Strong interoperability with enterprise systems |
| System Complexity | High | Designed for large-scale, complex operations |
| Innovation Readiness | AI/ML-ready via Polar platform | Supports future digital transformation initiatives |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic considerations from IDBS E-WorkBook’s positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Regulatory Requirements | Ideal for highly regulated pharmaceutical environments |
| Scalability Needs | Suitable for large, multi-site enterprise operations |
| Implementation Resources | Requires significant IT and validation investment |
| Innovation Readiness | Supports long-term AI and data-driven research strategies |
IDBS E-WorkBook remains one of the most authoritative and reliable ELN platforms in 2026, particularly for enterprise-scale organizations operating in regulated industries. Its combination of deep compliance capabilities, extensive integration options, and proven performance makes it a cornerstone solution for global pharmaceutical research, even as organizations balance these strengths against the complexity and resource demands of implementation.
7. Revvity Signals Notebook
Within the global Electronic Lab Notebook (ELN) software landscape in 2026, Revvity Signals Notebook has established itself as a leading cloud-first platform, particularly recognized for its strong capabilities in chemistry-focused research environments. Formerly part of PerkinElmer, the platform benefits from its integration within the broader Revvity Signals ecosystem, enabling advanced analytics, visualization, and scientific data management.
Signals Notebook is especially well-suited for early-stage research teams and organizations operating in chemistry-intensive domains, where data visualization, molecular modeling, and analytical insights are critical to accelerating discovery. Its modern interface and intuitive design make it appealing to researchers seeking a balance between usability and advanced functionality.
Core Platform Capabilities and Visualization Strengths
Signals Notebook differentiates itself through its emphasis on visual data representation and chemistry-specific tools, enabling researchers to gain deeper insights into experimental results.
| Core Capability | Functional Description | Strategic Value for Chemistry Research |
|---|---|---|
| Cloud-First Architecture | Fully cloud-based platform with scalable infrastructure | Enhances accessibility and collaboration across teams |
| Advanced Data Visualization | Graphical representation of experimental and chemical data | Improves interpretation and decision-making |
| Chemistry-Focused Tools | Supports chemical structures, reactions, and analytical workflows | Optimizes workflows for chemistry-driven research |
| Modern User Interface | Clean and intuitive design | Reduces friction and enhances user experience |
These capabilities position Signals Notebook as a highly effective platform for research environments that prioritize visual analytics and chemistry-specific workflows.
Integration Within the Revvity Signals Ecosystem
A key strength of Signals Notebook lies in its deep integration with the broader Revvity Signals platform, which provides access to a suite of advanced scientific tools and analytics.
| Ecosystem Component | Functionality | Strategic Benefit |
|---|---|---|
| Signals Analytics | High-end data analysis tools | Enables advanced interpretation of complex datasets |
| Signals Data Management | Centralized storage and organization of scientific data | Improves data governance and accessibility |
| Signals Collaboration | Cross-team sharing and communication tools | Enhances collaboration in distributed research environments |
This ecosystem-driven approach allows organizations to scale their capabilities by leveraging multiple interconnected tools.
User Profile and Ideal Use Cases
Signals Notebook is particularly well-aligned with organizations that require advanced chemistry tools and have the resources to support premium software investments.
| User Segment | Suitability Level | Key Insight |
|---|---|---|
| Large Enterprises | High | Ideal for organizations with complex chemistry workflows |
| Mid-Market Organizations | Moderate | Suitable but may face budget constraints |
| Small Laboratories | Low | Pricing limits accessibility for smaller teams |
| Early-Stage Research | High | Strong fit for chemistry-focused innovation environments |
This distribution highlights its positioning as a premium solution tailored for high-value research environments.
Performance Metrics and Platform Characteristics
Signals Notebook maintains strong performance in usability and compliance, although certain operational considerations impact its overall positioning.
| Performance Metric | Signals Notebook Evaluation | Strategic Interpretation |
|---|---|---|
| User Interface | Clean and modern | Enhances usability and user satisfaction |
| Implementation | Moderate complexity | Requires structured onboarding and configuration |
| Primary Use Case | High-end chemistry research | Strong specialization in chemistry workflows |
| Compliance Capability | 21 CFR Part 11 compliant | Suitable for regulated environments |
Limitations and Innovation Gaps
Despite its strengths, Signals Notebook faces several limitations that may affect its competitiveness in a rapidly evolving ELN market.
| Limitation Area | Description | Impact on Organizations |
|---|---|---|
| AI Capabilities | Lacks native AI-driven automation | Limits advanced predictive analytics and workflow automation |
| Innovation Pace | Slower development compared to newer platforms | May lag behind in adopting emerging technologies |
| Pricing Accessibility | High cost structure | Restricts adoption among small and mid-sized labs |
| LIMS Integration | No built-in LIMS for QC or batch testing | Requires additional systems for full workflow coverage |
These limitations highlight the need for organizations to evaluate whether additional tools or integrations are required to achieve a complete laboratory ecosystem.
Pricing Positioning and Market Accessibility
Signals Notebook is positioned as a premium ELN solution, reflecting its advanced capabilities and integration within a broader scientific software ecosystem.
| Pricing Dimension | Signals Notebook Positioning | Market Implication |
|---|---|---|
| Cost Level | High | Suitable for organizations with larger budgets |
| Value Proposition | Advanced visualization and chemistry tools | Strong ROI for specialized research environments |
| Accessibility | Limited for smaller labs | May require budget justification for adoption |
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, Signals Notebook is positioned as a premium, chemistry-focused platform that excels in visualization and analytical capabilities.
| Competitive Dimension | Signals Notebook Positioning | Market Advantage |
|---|---|---|
| Core Focus | Chemistry and early-stage research | Deep specialization in chemical workflows |
| Visualization Capability | Very high | Industry-leading graphical data representation |
| Ecosystem Integration | Strong | Access to broader Revvity Signals tools |
| AI Innovation | Limited | Lags behind AI-first ELN platforms |
| Workflow Completeness | Partial | Requires additional tools for full lab management |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic considerations from Signals Notebook’s positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Research Focus | Ideal for chemistry-intensive laboratories |
| Budget Considerations | Requires higher investment compared to many alternatives |
| Integration Needs | May require additional systems for full workflow coverage |
| Innovation Requirements | Limited AI capabilities compared to newer platforms |
Revvity Signals Notebook remains a strong contender within the global ELN market in 2026, particularly for organizations prioritizing advanced chemistry tools and data visualization capabilities. While its premium positioning and ecosystem integration provide significant value for large-scale research environments, its limitations in AI automation and workflow completeness require careful consideration when compared to newer, more agile ELN platforms.
8. Dotmatics ELN
In the global Electronic Lab Notebook (ELN) software landscape in 2026, Dotmatics has established itself as a powerful, enterprise-grade platform that bridges the gap between scientific informatics and laboratory data management. Designed to support large-scale, data-intensive research environments, Dotmatics is particularly well-suited for organizations operating in chemistry, material science, and multidisciplinary R&D domains.
Unlike lightweight ELN tools focused primarily on documentation, Dotmatics delivers a comprehensive scientific data ecosystem that integrates ELN capabilities with advanced informatics, analytics, and data management. This makes it a preferred solution for organizations handling high volumes of complex experimental data across multiple research functions.
Core Platform Capabilities and Scientific Informatics Integration
Dotmatics distinguishes itself through its ability to unify diverse scientific data streams into a cohesive and scalable platform, enabling researchers to manage, analyze, and interpret data more effectively.
| Core Capability | Functional Description | Strategic Value for Research Organizations |
|---|---|---|
| ELN Functionality | Centralized experimental documentation and data capture | Ensures data traceability and collaboration |
| Scientific Informatics | Advanced tools for data analysis and interpretation | Enables deeper scientific insights |
| Multidisciplinary Support | Handles chemical, biological, and analytical data | Supports cross-functional research environments |
| Data Integration | Connects multiple data sources and systems | Reduces silos and improves data consistency |
This integrated approach allows organizations to move beyond simple record-keeping toward a more holistic, data-driven research model.
Organizational Scale and R&D Investment
Dotmatics’ global presence and strong investment in research and development reinforce its position as a leader in scientific software innovation.
| Organizational Metric | Quantitative Value | Strategic Insight |
|---|---|---|
| Global Workforce | Over 800 employees | Significant organizational scale and expertise |
| R&D Workforce Share | 65% | Strong focus on continuous innovation and product development |
| Countries Served | 125 | Extensive global reach across research markets |
This level of investment enables Dotmatics to continuously enhance its platform capabilities and maintain competitiveness in a rapidly evolving market.
Global Adoption and Platform Scale
Dotmatics demonstrates substantial global adoption, reflecting its ability to support enterprise-scale research operations.
| Adoption Metric | Quantitative Value | Market Implication |
|---|---|---|
| Global User Count | Over 2,000,000 users | High adoption across large-scale organizations |
| Global Customer Count | Over 10,000 customers | Broad client base across industries |
| Net Promoter Score (NPS) | 71 | Indicates strong customer satisfaction and loyalty |
These figures highlight Dotmatics’ credibility and effectiveness in delivering value to a wide range of scientific organizations.
Implementation Timeline and Scalability Considerations
Dotmatics is designed for large-scale deployments, which is reflected in its implementation process and scalability capabilities.
| Implementation Factor | Dotmatics Performance | Strategic Consideration |
|---|---|---|
| Implementation Timeline | Several months to one year | Requires long-term planning and resource allocation |
| Scalability | Very high | Supports enterprise-level, high-volume data environments |
| Customization Depth | Extensive | Allows tailored workflows but increases complexity |
While the platform offers significant flexibility, its implementation demands careful planning and strong technical support.
Strengths in High-Volume and Multidisciplinary Research
Dotmatics excels in environments where large datasets and complex workflows are the norm, particularly in chemistry and material science.
| Strength Area | Description | Strategic Advantage |
|---|---|---|
| High-Volume Data Handling | Processes large datasets efficiently | Ideal for data-intensive research environments |
| Cross-Disciplinary Support | Integrates multiple scientific domains | Enables collaborative, multidisciplinary innovation |
| Advanced Analytics | Provides deep insights into experimental data | Supports informed decision-making |
These strengths make Dotmatics a compelling choice for organizations requiring robust data infrastructure.
Limitations and User Experience Challenges
Despite its extensive capabilities, Dotmatics presents several challenges that may impact user adoption and operational efficiency.
| Limitation Area | Description | Impact on Users |
|---|---|---|
| Customization Complexity | Requires significant effort to configure workflows | Increases implementation time and technical dependency |
| User Interface | Considered less modern compared to newer platforms | May reduce user engagement and ease of use |
| Implementation Duration | Long deployment cycles | Delays time-to-value for organizations |
These factors highlight the trade-off between advanced functionality and usability.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, Dotmatics is positioned as a high-capacity, informatics-driven platform designed for enterprise-scale scientific research.
| Competitive Dimension | Dotmatics Positioning | Market Advantage |
|---|---|---|
| Core Focus | Scientific informatics and ELN integration | Strong alignment with data-intensive research environments |
| Scalability | Very high | Supports large-scale, global operations |
| Feature Depth | Extensive | Comprehensive functionality across scientific workflows |
| Ease of Use | Moderate to low | Requires training and technical expertise |
| Implementation Speed | Slow | Long-term deployment approach |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic considerations from Dotmatics’ positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Data Volume Requirements | Ideal for high-volume, data-intensive environments |
| Customization Needs | Offers flexibility but requires technical resources |
| Implementation Timeline | Suitable for long-term transformation projects |
| User Experience Priorities | May require trade-offs compared to modern SaaS platforms |
Dotmatics stands out as a highly scalable and powerful ELN platform in 2026, particularly for organizations operating at the intersection of scientific informatics and large-scale data management. Its ability to support complex, multidisciplinary research workflows makes it a strong contender in the global ELN market, even as organizations carefully weigh its implementation complexity and user experience against more agile, cloud-native alternatives.
9. SciSure (eLabNext)
In the global Electronic Lab Notebook (ELN) software market in 2026, SciSure—previously known as eLabNext—has emerged as a highly flexible and accessible “Digital Lab Platform” tailored for research organizations seeking a balance between functionality, usability, and scalability. Designed to integrate ELN, LIMS, and sample tracking into a unified system, SciSure is particularly popular among biotechnology startups, academic institutions, and laboratories transitioning from paper-based workflows.
Its modular architecture allows organizations to adopt and expand functionalities progressively, making it a practical solution for teams that require adaptability without the complexity of fully enterprise-grade systems. This flexibility, combined with strong community support, positions SciSure as a versatile platform for general research and development environments.
Core Platform Capabilities and Modular Architecture
SciSure delivers a comprehensive set of laboratory management tools while maintaining a user-friendly and customizable structure. Its modular design enables laboratories to tailor the platform to their specific operational needs.
| Core Capability | Functional Description | Strategic Value for R&D Teams |
|---|---|---|
| Electronic Lab Notebook | Centralized experiment documentation and record-keeping | Improves data organization and reproducibility |
| Sample Tracking | Monitors samples with barcode and labeling systems | Enhances traceability and reduces manual errors |
| LIMS Integration (Basic) | Supports workflow and sample management | Provides foundational lab management capabilities |
| Protocol Management | Intuitive creation and reuse of experimental procedures | Standardizes workflows and improves efficiency |
| Modular System Design | Expandable features based on organizational needs | Enables scalable adoption without overcomplication |
This integrated yet flexible approach makes SciSure particularly suitable for laboratories that require a practical and adaptable digital infrastructure.
User Adoption and Market Distribution
SciSure demonstrates strong adoption among smaller organizations and academic environments, reflecting its accessibility and ease of use.
| User Segment | Percentage Share | Key Insight |
|---|---|---|
| Small Businesses | 71% | High adoption among startups and early-stage research teams |
| Mid-Market Organizations | 15% | Growing usage among scaling laboratories |
| Other Segments | 14% | Includes academic institutions and niche research groups |
This distribution highlights SciSure’s positioning as an entry-to-mid-level platform that supports organizations during early growth phases.
Usability and Satisfaction Metrics
SciSure is widely recognized for its ease of use and intuitive interface, making it one of the more accessible ELN platforms in the market.
| Satisfaction Metric | SciSure Performance | Market Interpretation |
|---|---|---|
| Overall Rating | 4.2 / 5.0 | Strong user satisfaction across diverse user groups |
| Total Reviews | 296 | High level of user feedback and validation |
| Ease of Use Rating | 8.8 | Ranked among the easiest ELN platforms |
| Ease of Use Ranking | 4th easiest ELN | Reflects strong usability compared to competitors |
These metrics reinforce SciSure’s reputation as a user-friendly solution that minimizes onboarding challenges.
Strengths in Workflow Efficiency and Sample Management
SciSure’s capabilities are particularly effective in simplifying day-to-day laboratory operations, especially for research teams focused on documentation and sample handling.
| Strength Area | Description | Strategic Advantage |
|---|---|---|
| Protocol Management | Easy creation and reuse of experiment templates | Improves consistency and reduces setup time |
| Barcode and Labeling | Integrated sample tracking system | Enhances accuracy and traceability |
| Workflow Simplicity | Intuitive interface and navigation | Reduces training requirements |
| Community Support | Active user and developer community | Facilitates knowledge sharing and platform improvement |
These strengths make SciSure a practical solution for laboratories prioritizing efficiency and simplicity.
Limitations and Functional Gaps
While SciSure offers strong flexibility and usability, it presents certain limitations that may affect its suitability for more complex or regulated environments.
| Limitation Area | Description | Impact on Organizations |
|---|---|---|
| Advanced LIMS Functionality | Limited support for manufacturing and QC stability workflows | Not ideal for highly regulated production environments |
| Offline Capability | No offline mode available | Limits usability in environments with restricted connectivity |
| Enterprise Scalability | Less robust for large-scale operations | May require migration as organizations grow |
These limitations highlight its positioning as a flexible, mid-tier platform rather than a full enterprise solution.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, SciSure is positioned as a flexible, modular platform that prioritizes usability and adaptability over complexity.
| Competitive Dimension | SciSure Positioning | Market Advantage |
|---|---|---|
| Core Focus | General R&D and academic research | Strong alignment with early-stage and flexible environments |
| Ease of Use | Very high | Minimal training and fast adoption |
| Feature Complexity | Moderate | Balanced functionality without overwhelming users |
| Scalability | Moderate | Suitable for growing but not enterprise-scale labs |
| Customization | Flexible | Modular design supports tailored workflows |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic considerations from SciSure’s positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Ease of Adoption | Ideal for labs transitioning from paper-based systems |
| Flexibility Needs | Modular design supports evolving research requirements |
| Budget Sensitivity | More accessible compared to enterprise-grade platforms |
| Advanced Workflow Needs | May require alternative solutions for manufacturing or QC |
SciSure (eLabNext) stands out as a flexible and user-friendly digital lab platform in 2026, offering strong value for academic institutions, biotech startups, and research teams seeking efficient documentation and sample tracking. Its modular design and ease of use make it an attractive option for growing laboratories, even as organizations with more complex regulatory or operational requirements may need to consider more advanced enterprise solutions.
10. LabWare ELN
In the global Electronic Lab Notebook (ELN) and Laboratory Information Management System (LIMS) market in 2026, LabWare is widely recognized as one of the most established and extensively deployed platforms, serving more than 14,000 laboratories across 125 countries. It has become the preferred solution for large, regulated organizations—particularly in pharmaceutical quality assurance (QA), quality control (QC), and industrial testing environments—where precision, compliance, and scalability are critical.
LabWare’s strength lies in its deeply integrated ELN and LIMS ecosystem, which supports highly structured workflows, advanced laboratory automation, and strict regulatory adherence. Its long-standing presence in the market and proven track record make it a cornerstone platform for enterprises operating in compliance-heavy industries.
Core Platform Capabilities and Compliance-Driven Architecture
LabWare is designed to meet the complex demands of regulated laboratories, offering a comprehensive suite of tools that ensure data integrity, traceability, and workflow standardization.
| Core Capability | Functional Description | Strategic Value for Regulated Enterprises |
|---|---|---|
| Integrated ELN + LIMS | Unified platform for experiment documentation and workflow control | Eliminates system silos and improves data consistency |
| Laboratory Automation | Supports complex, automated lab processes | Enhances operational efficiency and throughput |
| Data Integrity Framework | Ensures secure and validated data handling | Supports regulatory compliance and audit readiness |
| Workflow Standardization | Predefined and customizable workflows | Improves reproducibility and operational control |
These capabilities make LabWare particularly suitable for organizations that require strict adherence to global regulatory standards and high levels of operational precision.
Global Adoption and Market Leadership
LabWare’s extensive deployment across industries highlights its dominance in the enterprise ELN and LIMS space.
| Adoption Metric | Quantitative Value | Strategic Insight |
|---|---|---|
| Laboratories Served | Over 14,000 | Broad global adoption across multiple industries |
| Countries Covered | 125 | Strong international presence |
| Primary Use Case | Pharmaceutical QA/QC and regulated labs | Focus on compliance-intensive environments |
This scale reinforces LabWare’s reputation as a trusted platform for mission-critical laboratory operations.
Performance Metrics and User Satisfaction
LabWare consistently achieves high performance ratings in key areas related to data management and security, reflecting its strength in enterprise environments.
| Performance Metric | LabWare Score | Category Average | Performance Insight |
|---|---|---|---|
| Data Storage | 9.4 | 8.8 | Exceptional capability in managing large datasets |
| Data Access Authorization | 9.3 | 8.8 | Strong control over user permissions and data security |
| Protocol Templates | 8.8 | 8.8 | Meets industry standards for workflow standardization |
| Ease of Use | 8.3 | 8.8 | Lower usability due to system complexity |
| Customer Satisfaction | 98% | — | Indicates strong long-term user confidence |
These metrics demonstrate LabWare’s strength in data governance and reliability, even as usability remains a challenge for some users.
Scalability and Automation Capabilities
LabWare is engineered to support large-scale, automated laboratory environments, making it highly suitable for enterprises managing complex workflows and high data volumes.
| Capability Area | Description | Strategic Advantage |
|---|---|---|
| High-Volume Data Handling | Processes large and complex datasets efficiently | Supports enterprise-scale operations |
| Automation Integration | Enables end-to-end laboratory automation | Reduces manual intervention and improves throughput |
| Workflow Customization | Tailors processes to specific regulatory and operational needs | Enhances flexibility within structured environments |
These features position LabWare as a robust platform for organizations seeking long-term scalability and operational control.
Implementation Complexity and Resource Requirements
Despite its extensive capabilities, LabWare is often associated with significant implementation complexity and resource demands.
| Implementation Factor | LabWare Performance | Impact on Organizations |
|---|---|---|
| Implementation Timeline | Approximately 7 months | Requires long-term planning and phased deployment |
| ROI Period | Around 44 months | Longer time-to-value compared to modern SaaS platforms |
| Technical Requirements | High | Requires specialized IT expertise and infrastructure |
| Configuration Approach | Proprietary scripting | Limits flexibility and increases dependency on experts |
| Setup Support | Certified consultants needed | Adds to implementation cost and complexity |
These factors highlight the trade-off between enterprise-grade functionality and operational agility.
Limitations and Usability Considerations
While LabWare excels in compliance and scalability, it presents several challenges that may affect user experience and adaptability.
| Limitation Area | Description | Impact on Users |
|---|---|---|
| System Rigidity | Highly structured workflows | Limits flexibility for dynamic research environments |
| Learning Curve | Complex interface and functionality | Requires extensive user training |
| Implementation Overhead | Resource-intensive deployment | Increases upfront investment and time commitment |
These limitations suggest that LabWare is best suited for organizations that prioritize stability and compliance over rapid adaptability.
Strategic Positioning in the ELN Market in 2026
Within the Top 10 Electronic Lab Notebook Software in the world in 2026, LabWare is positioned as a leading enterprise solution for regulated laboratories requiring high levels of compliance and automation.
| Competitive Dimension | LabWare Positioning | Market Advantage |
|---|---|---|
| Core Focus | Enterprise QA/QC and regulated labs | Strong alignment with compliance-heavy industries |
| Compliance Strength | Very high | Industry-leading regulatory support |
| Scalability | Very high | Supports global, multi-site laboratory operations |
| Ease of Use | Moderate to low | Complex system requiring training |
| Implementation Speed | Slow | Long deployment and ROI timelines |
Key Insights for ELN Buyers
Organizations evaluating ELN platforms in 2026 can derive several strategic considerations from LabWare’s positioning:
| Decision Factor | Strategic Insight |
|---|---|
| Compliance Requirements | Ideal for highly regulated environments |
| Scalability Needs | Suitable for large, global laboratory networks |
| Implementation Resources | Requires significant investment in time and expertise |
| Usability Priorities | May not suit teams seeking simplicity and rapid adoption |
LabWare ELN remains one of the most authoritative and widely deployed platforms in the global ELN market in 2026. Its unmatched strength in compliance, automation, and scalability makes it a critical solution for enterprise laboratories, particularly in regulated industries. However, organizations must carefully evaluate its complexity, implementation requirements, and long-term investment when compared to more agile, modern alternatives.
The Global Electronic Lab Notebook Landscape in 2026: Market Evolution and Technological Convergence
The scientific research environment in 2026 is undergoing a profound transformation, characterized by a shift from passive data documentation to active, intelligence-driven research ecosystems. Electronic Lab Notebooks (ELNs), once considered simple digital repositories, have evolved into central operational platforms that orchestrate the entire laboratory workflow.
Modern ELNs now function as the core infrastructure of the laboratory, integrating artificial intelligence, cloud-native computing, and advanced compliance frameworks. These systems are designed to manage the immense scale of data generated by the life sciences sector, which has reached an estimated 40 exabytes annually. This exponential growth in data volume has necessitated a redefinition of how research is conducted, with ELNs acting not only as record-keeping tools but as intelligent systems that assist in hypothesis generation, data interpretation, and decision-making.
A key trend shaping the industry is the convergence of ELN, Laboratory Information Management Systems (LIMS), and Scientific Data Management Systems (SDMS) into unified platforms. These integrated environments, often referred to as Lab Operating Systems (LabOS), represent the next generation of laboratory infrastructure. By consolidating data management, workflow automation, and analytics into a single ecosystem, LabOS platforms enable research and development organizations to operate with greater efficiency, scalability, and regulatory alignment.
Market Dynamics and Economic Outlook in 2026
The global Electronic Lab Notebook market in 2026 reflects strong growth momentum, driven by increased demand for digital transformation in scientific research and heightened regulatory requirements.
| Metric | 2025 Value | 2026 Estimated Value | Long-Term Projection | CAGR (2026–2035) |
|---|---|---|---|---|
| Global ELN Market (USD) | 750.82 Million | 805.33 Million | 1,513.26 Million (2035) | 7.26% |
| U.S. ELN Market (USD) | 220 Million | 235 Million | 410 Million (2035) | 6.81% |
| Enterprise Informatics (USD) | 6.0 Billion | 6.6 Billion | 19.8 Billion (2036) | 7.00% |
| Life Sciences Market Share (%) | 43% | 46.12% | Leading Segment | — |
The ELN market has expanded from USD 750.82 million in 2025 to an estimated USD 805.33 million in 2026, reflecting a steady upward trajectory. Forecasts indicate that the market will surpass USD 1.5 billion by 2035, supported by a compound annual growth rate of 7.26%.
However, when viewed within the broader context of enterprise laboratory informatics—which includes LIMS, SDMS, and integrated data platforms—the market opportunity is significantly larger. In 2026, this expanded segment is valued at approximately USD 6.6 billion and is projected to reach nearly USD 19.8 billion by 2036.
This divergence in market size estimates highlights the increasing overlap between traditional ELN tools and comprehensive laboratory platforms. As vendors expand their capabilities to include sample tracking, instrument integration, and quality management, the distinction between standalone ELNs and full laboratory ecosystems continues to diminish.
Key Market Drivers and Industry Adoption Patterns
The economic structure of the ELN market in 2026 is heavily influenced by large pharmaceutical and biotechnology organizations, which together account for nearly half of total industry revenue.
| Industry Segment | Market Contribution | Growth Trend | Strategic Importance |
|---|---|---|---|
| Pharmaceutical & Biotechnology | 48% | Stable and dominant | Drives demand for compliance and large-scale data systems |
| Contract Research Organizations | Rapid growth | 8.25% CAGR | Increasing demand for outsourced research capabilities |
| Academic & Research Institutions | Moderate | Steady adoption | Focus on affordability and usability |
| Industrial & Chemical Sectors | Expanding | Moderate growth | Emphasis on process optimization and data integration |
While pharmaceutical and biotech firms remain the largest contributors to market revenue, the fastest-growing segment is the Contract Research Organization (CRO) sector. With a projected CAGR of 8.25%, CROs are benefiting from the increasing trend toward outsourced research and development.
This shift toward collaborative and distributed R&D models has introduced new requirements for ELN platforms, particularly the ability to share secure, validated, and compliant data across organizational boundaries in real time. As a result, interoperability, cloud accessibility, and data standardization have become critical competitive differentiators among ELN vendors.
Technological Convergence and the Rise of Lab Operating Systems
The convergence of multiple laboratory technologies into unified platforms is one of the most significant developments in the ELN market in 2026.
| Technology Component | Traditional Role | Integrated Role in LabOS |
|---|---|---|
| ELN | Experimental documentation | Central data hub and collaboration platform |
| LIMS | Sample and workflow management | Operational backbone for laboratory processes |
| SDMS | Data storage and organization | Scalable data infrastructure and analytics layer |
| AI and Machine Learning | Limited or external tools | Embedded intelligence for predictive insights |
Lab Operating Systems (LabOS) combine these components into a unified architecture, enabling laboratories to operate more efficiently and intelligently. These platforms are designed to handle complex workflows, automate repetitive tasks, and provide real-time insights into experimental outcomes.
The integration of artificial intelligence further enhances these systems by enabling predictive analytics, automated protocol generation, and intelligent data interpretation. This shift transforms the ELN from a passive record-keeping tool into an active participant in the research process.
Strategic Implications for R&D Organizations
The evolution of ELN platforms in 2026 presents both opportunities and challenges for research organizations.
| Strategic Factor | Implication for Organizations |
|---|---|
| Data Volume Growth | Requires scalable, cloud-native infrastructure |
| Regulatory Complexity | Necessitates robust compliance and audit capabilities |
| Collaboration Needs | Demands secure, real-time data sharing across teams |
| Technology Integration | Encourages adoption of unified LabOS platforms |
| AI Adoption | Enables faster discovery and improved decision-making |
Organizations that successfully leverage these advancements can achieve significant gains in productivity, data accuracy, and innovation speed. Conversely, those relying on legacy systems may face increasing inefficiencies and competitive disadvantages.
Conclusion: The Future Direction of ELN Platforms
The Electronic Lab Notebook market in 2026 is no longer defined solely by digital record-keeping. Instead, it represents a critical component of a broader digital transformation within scientific research.
As ELNs continue to evolve into intelligent, integrated platforms, their role within the laboratory will expand further—enabling not only the management of data but also the acceleration of discovery itself. The convergence of technologies into Lab Operating Systems signals a future where laboratories operate as fully connected, data-driven ecosystems, capable of responding dynamically to the complexities of modern research.
This transformation underscores the importance of selecting ELN platforms that are not only functionally robust but also adaptable to the rapidly changing demands of scientific innovation.
Regional Market Analysis and Growth Drivers in the Electronic Lab Notebook Market (2026)
The global Electronic Lab Notebook (ELN) market in 2026 is shaped by distinct regional dynamics, each driven by regulatory frameworks, research investment levels, and digital transformation priorities. While North America continues to dominate in terms of market share and innovation leadership, emerging economies in the Asia-Pacific region are rapidly accelerating adoption, reshaping the global competitive landscape.
At the same time, Europe is undergoing significant modernization efforts to align with stringent regulatory requirements, further contributing to global market expansion. Across all regions, the transition toward cloud-native laboratory ecosystems remains the most influential technological driver.
Regional Market Share and Growth Outlook
The distribution of market share and projected growth rates highlights the shifting balance of global influence within the ELN sector.
| Region | 2026 Market Share | Forecasted CAGR | Primary Growth Drivers |
|---|---|---|---|
| North America | 40.50% | 6.81% | High R&D spending; strong biotech ecosystem; policy support |
| Asia-Pacific | 20.33% | 8.71% | Regulatory reforms; rapid digital transformation |
| Europe | 21.44% | 7.75% | Compliance requirements; legacy system modernization |
| China | — | 10.20% | Pharmaceutical and CRO sector modernization |
| India | — | 10.00% | Manufacturing compliance and regulatory upgrades |
This regional breakdown illustrates a mature yet evolving market, where growth is increasingly driven by regulatory enforcement and digital adoption in emerging economies.
North America: Market Leadership Through Innovation and Investment
North America remains the largest and most mature ELN market in 2026, accounting for over 40% of global market share. This dominance is largely attributed to the substantial research and development investments made by the United States, particularly within the biotechnology and pharmaceutical sectors.
| Key Factor | Description | Strategic Impact |
|---|---|---|
| R&D Investment | High expenditure by biotech and pharma companies | Drives demand for advanced ELN platforms |
| Vendor Presence | Headquarters of leading ELN providers | Accelerates innovation and ecosystem development |
| Regulatory Environment | Strong policy frameworks (e.g., NIH guidelines) | Encourages adoption of compliant digital systems |
The region’s leadership is further reinforced by its early adoption of AI-driven laboratory platforms and cloud-based infrastructures.
Asia-Pacific: The Fastest-Growing Regional Market
The Asia-Pacific (APAC) region is emerging as the fastest-growing market for ELN solutions, with a projected CAGR of 8.71%. This growth is primarily driven by regulatory transformations and increasing investments in pharmaceutical manufacturing and research.
| Growth Driver | Description | Regional Impact |
|---|---|---|
| Regulatory Reforms | Implementation of NMPA regulations in China and Schedule M updates in India | Forces transition from paper to digital systems |
| Pharmaceutical Expansion | Growth of domestic pharma and CRO industries | Increases demand for scalable ELN and LIMS solutions |
| Digital Transformation | Government and private sector initiatives | Accelerates adoption of cloud-based laboratory platforms |
Key High-Growth Markets Within APAC
| Country | Forecasted CAGR | Key Growth Catalyst |
|---|---|---|
| China | 10.20% | Pharma and CRO modernization |
| India | 10.00% | Manufacturing compliance and regulatory alignment |
These developments position APAC as a critical growth engine for ELN vendors, particularly those offering compliant, scalable, and cost-effective solutions.
Europe: Compliance-Driven Modernization
European markets are undergoing a significant transformation, driven by stringent regulatory requirements and the need to modernize legacy laboratory systems.
Germany leads the region with a projected CAGR of 9.4%, reflecting strong industrial and pharmaceutical activity.
| Key Factor | Description | Strategic Impact |
|---|---|---|
| Annex 11 Compliance | Regulatory requirement for electronic records and signatures | Drives adoption of validated ELN systems |
| ISO and REACH Standards | Environmental and quality compliance frameworks | Encourages digital traceability and data integrity |
| Legacy System Upgrades | Replacement of outdated informatics infrastructure | Creates demand for modern, integrated platforms |
Europe’s focus on compliance and sustainability is shaping the adoption of robust, audit-ready ELN solutions.
Technological Driver: The Rise of Cloud-Native ELN Platforms
Across all regions, the transition toward cloud-native ELN systems is the most significant technological trend influencing market growth.
| Deployment Model | Market Share (2026) | Key Advantages |
|---|---|---|
| Cloud-Based (Web-Hosted) | 68.12% | Lower costs; remote access; rapid deployment |
| On-Premise | 31.88% | Greater control; legacy system compatibility |
Cloud-based deployments now dominate the market, accounting for over two-thirds of total adoption. This shift is driven by several key factors:
- Reduced infrastructure and maintenance costs
- Enhanced accessibility for remote and distributed teams
- Faster implementation and scalability
- Improved collaboration across global research networks
These advantages make cloud-native platforms particularly attractive for both large enterprises and emerging startups.
Strategic Implications for ELN Vendors and Buyers
The regional dynamics of the ELN market in 2026 present several important strategic considerations for both software vendors and research organizations.
| Strategic Factor | Implication |
|---|---|
| Regional Compliance Needs | Vendors must localize solutions to meet regulatory requirements |
| Emerging Market Growth | APAC represents the highest growth opportunity |
| Cloud Adoption Trends | Cloud-native platforms are becoming the industry standard |
| Competitive Differentiation | Integration, scalability, and compliance are key success factors |
Organizations that align their technology strategies with these regional and technological trends will be better positioned to capitalize on the evolving ELN market.
Conclusion: A Globally Diversifying and Rapidly Evolving Market
The Electronic Lab Notebook market in 2026 is no longer dominated solely by mature markets such as North America. Instead, it is becoming increasingly global, with Asia-Pacific emerging as a major growth driver and Europe reinforcing its position through compliance-led modernization.
At the same time, the widespread adoption of cloud-native platforms is reshaping how laboratories operate, enabling greater flexibility, scalability, and collaboration.
As regulatory pressures intensify and research becomes more data-driven, the ability to deploy compliant, integrated, and cloud-based ELN solutions will be a defining factor in the success of both vendors and research organizations worldwide.
The Technological Vanguard: AI and the Emergence of the Active Laboratory in 2026
In 2026, the Electronic Lab Notebook (ELN) has undergone a fundamental transformation driven by the widespread integration of Generative Artificial Intelligence (GenAI) and Large Language Models (LLMs). This evolution has moved laboratory systems beyond passive data storage toward a state of “Active Maturity,” where ELNs function as intelligent collaborators within the scientific workflow.
Historically, one of the most significant bottlenecks in research productivity has been the disconnect between data capture and data interpretation. While ELNs successfully digitized experimental documentation, they failed to assist scientists in extracting meaning from the data. Approximately 81% of researchers reported that their ELN systems offered no support in interpreting experimental results, creating a paradox where increased data generation actually slowed decision-making due to fragmented analysis across multiple tools.
By 2026, this limitation has been largely resolved. Modern ELNs now embed science-aware AI directly into the research workflow, enabling real-time interpretation, contextual insights, and automated reasoning. As a result, the role of the ELN has shifted from a passive digital repository to an active research partner that supports scientists throughout the discovery process.
From Passive Systems to Active Research Partners
The transition from traditional ELNs to AI-powered platforms represents a fundamental shift in how laboratories operate.
| System Type | Traditional ELNs (Pre-2026) | AI-Driven ELNs (2026) |
|---|---|---|
| Core Function | Data recording and storage | Data interpretation and decision support |
| Workflow Role | Passive documentation tool | Active research assistant |
| Data Utilization | Static records | Dynamic, insight-driven analysis |
| Integration Level | Fragmented tools | Unified, AI-enabled ecosystem |
This transformation has enabled laboratories to significantly reduce the time between experimentation and actionable insights, thereby accelerating innovation cycles.
AI Adoption and the End of Shadow AI
The adoption of AI within laboratory environments has reached near-universal levels in 2026, fundamentally reshaping research practices.
| AI Adoption Metric | Value | Strategic Interpretation |
|---|---|---|
| Scientists Using AI | 97% | AI has become a standard tool in scientific research |
| Shadow AI Usage (Pre-2026) | 77% | Widespread use of external tools outside governance |
| Current State | Integrated AI | AI now embedded within compliant ELN ecosystems |
Previously, a significant proportion of researchers relied on external tools such as public AI platforms to assist with analysis, creating risks related to data security and compliance. This “Shadow AI” phenomenon has largely been eliminated as ELN vendors have incorporated secure, enterprise-grade AI capabilities directly into their platforms.
Key AI Capabilities and Adoption Rates in 2026
The most impactful AI applications in laboratory environments are those that integrate seamlessly into the daily workflows of scientists. These use cases focus on enhancing productivity without disrupting established research processes.
| AI Capability | Adoption Rate | Role in the 2026 Laboratory |
|---|---|---|
| Literature Review | 76% | Rapid synthesis and summarization of existing research |
| Protein Structure Prediction | 71% | Accelerates biologics and drug design processes |
| Scientific Reporting | 66% | Automates structured, compliant documentation |
| Target Identification | 58% | Identifies potential drug targets and biological pathways |
| Lead Optimization | 47% | Refines chemical compounds for improved efficacy |
| Generative Drug Design | 42% | Enables de novo molecule creation using AI models |
These applications are built on high-quality, structured datasets, allowing AI systems to deliver reliable insights while maintaining scientific integrity.
The Rise of AI “Co-Scientists” and Autonomous Research Support
Beyond task-specific automation, 2026 marks the emergence of AI systems that function as “co-scientists.” These advanced systems go beyond assisting with individual tasks and instead coordinate entire research workflows.
| AI Evolution Stage | Capability Description | Impact on Research |
|---|---|---|
| Task-Level Assistants | Perform isolated functions such as summarization or prediction | Improves efficiency in specific tasks |
| Workflow-Level AI | Integrates across multiple research processes | Enhances coordination and reduces fragmentation |
| AI Co-Scientists | End-to-end orchestration of experiments and decision-making | Transforms research into a proactive, intelligent system |
These AI co-scientists can:
- Anticipate resource requirements for experiments
- Suggest next steps based on historical data patterns
- Identify anomalies and propose plausible explanations
- Optimize experimental design in real time
This shift represents a move toward semi-autonomous research environments, where human scientists and AI systems collaborate closely to accelerate discovery.
The Importance of Structured Data Architecture
The effectiveness of AI-driven ELNs is fundamentally dependent on the quality and structure of the underlying data. Legacy systems that treated laboratory records as static digital pages are increasingly being replaced by data-centric architectures.
| Data Architecture Type | Legacy Systems | Modern AI-Driven Systems |
|---|---|---|
| Data Format | Unstructured or semi-structured | Fully structured and machine-readable |
| Analytical Capability | Limited | Advanced pattern recognition and prediction |
| Historical Insights | Difficult to extract | Easily accessible and actionable |
| AI Compatibility | Low | High |
Structured data enables AI systems to analyze trends across years of research, uncover hidden relationships, and generate predictive insights that were previously unattainable.
Strategic Implications for the Future of Scientific Research
The integration of AI into ELN platforms is reshaping the competitive landscape of scientific research in several key ways.
| Strategic Factor | Implication for Organizations |
|---|---|
| Decision-Making Speed | Accelerated through real-time AI insights |
| Data Utilization | Maximized through structured, analyzable datasets |
| Research Productivity | Increased via automation and intelligent recommendations |
| Compliance and Governance | Improved through integrated, secure AI systems |
| Innovation Potential | Enhanced through AI-driven discovery and experimentation |
Organizations that successfully adopt AI-enabled ELN platforms will gain a significant competitive advantage by reducing time-to-discovery and improving research outcomes.
Conclusion: The Active Lab as the New Standard
By 2026, the concept of the “Active Lab” has become the new standard in scientific research. ELNs are no longer passive tools for documentation but are evolving into intelligent systems that actively participate in the research process.
The convergence of AI, structured data, and integrated laboratory platforms has created an environment where experimentation, analysis, and decision-making occur seamlessly within a single ecosystem. This transformation not only enhances efficiency but also redefines the role of the scientist, enabling deeper insights and faster breakthroughs.
As the technology continues to mature, the laboratories that embrace AI-driven ELNs will be best positioned to lead the next wave of scientific innovation.
Regulatory Frameworks and Data Integrity in 2026
In 2026, regulatory compliance has become a foundational requirement for any organization operating within the life sciences and laboratory informatics ecosystem. The global compliance landscape is primarily shaped by stringent frameworks such as the FDA’s 21 CFR Part 11 in the United States and the European Union’s GMP Annex 11. These regulations define the standards under which electronic records and electronic signatures are considered legally equivalent to traditional paper-based documentation.
For modern laboratories, compliance is no longer treated as an optional feature or add-on capability. Instead, it is a prerequisite for market participation, particularly in pharmaceutical, biotechnology, and clinical research environments. ELN platforms must now be designed from the ground up to ensure data integrity, traceability, and auditability in accordance with these regulatory expectations.
Core Regulatory Requirements and Compliance Standards
The regulatory environment in 2026 emphasizes trust, reliability, and accountability in digital laboratory systems.
| Regulatory Framework | Region | Core Requirement | Strategic Impact |
|---|---|---|---|
| 21 CFR Part 11 | United States | Validates electronic records and signatures | Enables digital compliance in FDA-regulated industries |
| GMP Annex 11 | European Union | Governs computerized systems in regulated environments | Ensures integrity and traceability of laboratory data |
| GxP Standards | Global | Covers quality practices across manufacturing and research | Establishes baseline for compliance across industries |
These frameworks collectively ensure that laboratory data remains secure, verifiable, and suitable for regulatory inspection.
Audit Trails and the ALCOA+ Data Integrity Model
At the core of regulatory compliance in 2026 is the audit trail—a secure, system-generated record that captures every action performed within the ELN environment. This includes detailed logs of who performed an action, what changes were made, when they occurred, and why they were executed.
All leading ELN platforms adhere to the ALCOA+ model, which defines the essential principles of data integrity.
| ALCOA+ Attribute | Implementation in 2026 ELNs | Compliance Benefit |
|---|---|---|
| Attributable | Linked to unique user credentials; no shared access | Ensures accountability and traceability |
| Legible | Clear, human-readable records | Supports audit and review processes |
| Contemporaneous | Real-time timestamping using synchronized system clocks | Prevents backdating or manipulation of records |
| Original | Preservation of raw data from initial capture | Maintains authenticity of experimental data |
| Accurate | Automated calculations and direct instrument integration | Reduces human error and ensures data precision |
| Complete | Full data lifecycle captured without omissions | Ensures comprehensive documentation |
| Consistent | Standardized workflows and enforced protocols | Maintains uniformity across experiments |
| Enduring | Secure, long-term data storage | Protects data over extended regulatory timelines |
| Available | Accessible for audits and inspections | Facilitates regulatory review and compliance verification |
The ALCOA+ framework has become the global benchmark for evaluating the integrity and reliability of laboratory data systems.
The Emergence of Computer Software Assurance (CSA)
A significant development in 2026 is the adoption of the FDA’s Computer Software Assurance (CSA) approach, which represents a shift away from traditional, documentation-heavy validation processes.
| Validation Approach | Traditional Computer System Validation (CSV) | Computer Software Assurance (CSA) |
|---|---|---|
| Focus | Extensive documentation | Risk-based validation |
| Validation Scope | All system functions | Critical functions impacting quality and safety |
| Implementation Speed | Slow and resource-intensive | Faster and more efficient |
| Innovation Impact | Limits agility | Enables adoption of modern cloud solutions |
CSA allows organizations to prioritize validation efforts on high-risk areas that directly impact patient safety and product quality. This approach significantly reduces the burden of compliance while accelerating the adoption of cloud-native ELN platforms.
Blockchain and the Evolution of Data Provenance
In 2026, blockchain technology has emerged as a powerful tool for enhancing data integrity and traceability within laboratory environments. By providing a decentralized and tamper-evident record of data transactions, blockchain introduces an additional layer of trust to digital laboratory systems.
| Blockchain Capability | Description | Strategic Value |
|---|---|---|
| Immutable Data Records | Data entries are cryptographically secured and cannot be altered | Ensures authenticity and prevents data tampering |
| On-Chain Data Anchoring | Key data points are recorded on a distributed ledger | Enhances traceability across systems |
| Shared Trust Layer | Enables secure data sharing across organizations | Supports collaboration in multi-party research environments |
| IoMT Integration | Connects devices within the Internet of Medical Things | Ensures integrity of instrument-generated data |
This integration has led to the development of what is often referred to as an “AI and Data Provenance Fabric,” where both data and the processes used to generate it are fully traceable and verifiable.
Implications for AI and Predictive Analytics
The rise of AI-driven research in 2026 has placed greater emphasis on the quality and provenance of data used to train machine learning models.
| Data Requirement | Importance for AI Systems |
|---|---|
| Data Integrity | Ensures reliability of AI-generated insights |
| Data Provenance | Verifies origin and authenticity of training datasets |
| Structured Data Formats | Enables efficient analysis and pattern recognition |
| Compliance Alignment | Ensures AI outputs meet regulatory standards |
By leveraging blockchain and structured data architectures, organizations can ensure that AI models are trained on trusted, verifiable datasets. This is particularly critical in high-stakes applications such as drug discovery and clinical research, where data accuracy directly impacts outcomes.
Strategic Implications for Laboratory Organizations
The evolving regulatory and technological landscape in 2026 presents several strategic considerations for organizations adopting ELN platforms.
| Strategic Factor | Implication |
|---|---|
| Regulatory Compliance | Essential for market participation and audit readiness |
| Data Integrity | Critical for maintaining trust and scientific validity |
| Validation Approach | Shift toward CSA enables faster system adoption |
| Technology Integration | Blockchain enhances transparency and traceability |
| AI Readiness | Requires high-quality, structured, and verifiable data |
Organizations that successfully align their ELN strategies with these requirements will be better positioned to navigate regulatory complexities while leveraging advanced technologies for innovation.
Conclusion: Compliance as the Foundation of Digital Laboratories
In 2026, regulatory compliance and data integrity are no longer constraints but enablers of innovation within the laboratory environment. The integration of robust audit trails, adherence to the ALCOA+ model, adoption of CSA methodologies, and incorporation of blockchain technologies collectively establish a new standard for trustworthy scientific data systems.
As laboratories increasingly adopt AI-driven workflows and collaborative research models, the ability to ensure data authenticity, traceability, and compliance will be a defining factor in long-term success. ELN platforms that effectively combine regulatory rigor with technological innovation will serve as the backbone of the next generation of digital laboratories.
Comparative Analysis of Implementation Timelines and Return on Investment in 2026
In 2026, the evaluation of Electronic Lab Notebook (ELN) platforms is increasingly centered on a critical metric known as “Time to Science.” This concept refers to the duration between initial system deployment and the point at which researchers can fully leverage the platform to generate meaningful scientific outcomes.
As laboratories transition toward digital-first and AI-enabled environments, the speed of implementation and the time required to realize return on investment (ROI) have become decisive factors in software selection. Organizations are no longer willing to tolerate multi-year deployments without measurable productivity gains. Instead, they prioritize solutions that deliver rapid onboarding, immediate usability, and early-stage efficiency improvements.
Implementation Speed and ROI Benchmark Comparison
The following comparison highlights how leading ELN platforms differ in terms of deployment timelines, ROI realization, and implementation complexity.
| ELN Software | Time to Implement | Average ROI Time | Implementation Cost / Complexity |
|---|---|---|---|
| Genemod | Days | Immediate (AI-driven) | Low; cloud-native and rapid deployment |
| SciCord | Weeks | Rapid | Low; familiar spreadsheet-like interface |
| SciNote | Approximately 2 months | High for small teams | Low; simple cloud-based setup |
| Sapio LIMS | Around 6 months | Approximately 9 months | Moderate; customizable enterprise workflows |
| SampleManager | Around 7 months | Approximately 44 months | High; complex enterprise implementation |
| Benchling | Weeks to months | Variable | High; structured onboarding and enterprise setup |
This comparison illustrates a clear divide between modern, cloud-native platforms and traditional enterprise systems. Newer solutions emphasize speed, simplicity, and early value delivery, while legacy platforms often require longer deployment cycles due to their complexity and customization requirements.
The Shift Toward SaaS and Rapid Deployment Models
The widespread adoption of Software-as-a-Service (SaaS) delivery models has significantly transformed the ELN implementation landscape.
| Deployment Model | Key Characteristics | Impact on Organizations |
|---|---|---|
| Cloud-Based SaaS | Rapid deployment, minimal infrastructure requirements | Enables faster onboarding and reduced upfront costs |
| On-Premise Systems | Custom infrastructure and configuration | Leads to longer implementation timelines |
| Hybrid Models | Combination of cloud and on-premise | Balances flexibility with compliance needs |
Cloud-native ELN platforms now enable laboratories to go live within days or weeks, compared to several months for traditional systems. This acceleration is driven by pre-configured environments, automated updates, and reduced dependency on internal IT resources.
Measurable Productivity Gains and Operational Impact
One of the most compelling drivers of ELN adoption in 2026 is the ability to achieve immediate and measurable improvements in laboratory efficiency.
| Productivity Metric | Improvement After ELN Adoption | Strategic Benefit |
|---|---|---|
| Sample Search Time | Up to 90% reduction | Faster access to critical data |
| Documentation Efficiency | Significant improvement through automation | Reduces administrative burden on researchers |
| Collaboration Speed | Enhanced through cloud-based sharing | Improves cross-team coordination |
| Data Accessibility | Real-time access across locations | Supports global research operations |
These gains directly contribute to shorter research cycles and faster time-to-discovery.
Enterprise Implementation Challenges and Cost Considerations
While modern platforms emphasize speed and simplicity, enterprise-grade ELN systems continue to involve significant implementation complexity.
| Challenge Area | Description | Impact on Organizations |
|---|---|---|
| White-Glove Onboarding | High-touch implementation with dedicated support teams | Increases upfront costs |
| User Training | Extensive training for large research teams | Delays full system adoption |
| Data Structuring | Migration to structured data models | Requires careful planning and validation |
| Customization Requirements | Tailoring workflows to enterprise needs | Extends deployment timelines |
Platforms such as Benchling and IDBS often require substantial investment in onboarding and training, particularly when deploying across hundreds or thousands of users. This reflects the complexity of transitioning from unstructured or semi-structured systems to highly standardized, data-centric environments.
Strategic Trade-Off: Speed vs. Depth of Functionality
Organizations must carefully balance the trade-off between rapid implementation and depth of functionality when selecting an ELN platform.
| Decision Factor | Fast Deployment Platforms | Enterprise-Grade Platforms |
|---|---|---|
| Implementation Speed | Very fast (days to weeks) | Slow (months to over a year) |
| Initial ROI | Immediate or short-term | Long-term |
| Complexity | Low to moderate | High |
| Feature Depth | Moderate | Extensive |
| Scalability | High (for growth-stage labs) | Very high (for global enterprises) |
This distinction highlights the importance of aligning platform selection with organizational goals, resources, and long-term strategy.
Strategic Implications for ELN Buyers in 2026
The evolving implementation landscape presents several key considerations for organizations evaluating ELN platforms.
| Strategic Factor | Implication |
|---|---|
| Time to Science | Critical metric for evaluating platform effectiveness |
| Budget Constraints | Favors cloud-native, low-cost solutions |
| Organizational Scale | Determines suitability of enterprise-grade platforms |
| Training Requirements | Impacts speed of adoption and productivity gains |
| Long-Term Scalability | Influences future growth and system flexibility |
Organizations that prioritize rapid deployment and immediate productivity gains may favor modern SaaS platforms, while those with complex regulatory and operational requirements may opt for more comprehensive enterprise solutions despite longer implementation timelines.
Conclusion: Accelerating Scientific Outcomes Through Faster Implementation
In 2026, the success of an ELN platform is increasingly measured by how quickly it enables scientific progress. The concept of “Time to Science” has become a defining metric, reflecting the industry’s shift toward efficiency, agility, and data-driven decision-making.
The rise of cloud-native platforms has significantly reduced implementation timelines and accelerated ROI, enabling laboratories to achieve measurable benefits within weeks rather than months or years. At the same time, enterprise platforms continue to offer deep functionality and scalability, albeit with higher complexity and longer deployment cycles.
Ultimately, organizations must align their ELN strategy with their operational priorities—whether that is speed, scalability, compliance, or long-term innovation—to fully realize the transformative potential of digital laboratory systems.
The Path Toward 2030: Strategic Evolution of the Electronic Lab Notebook Market
As the Electronic Lab Notebook (ELN) market progresses through 2026 and approaches the end of the decade, the competitive landscape is increasingly defined by a “survival of the smartest” dynamic. Platforms that have remained static—functioning merely as digital record-keeping tools—are rapidly losing relevance. In contrast, AI-driven, cloud-native platforms that actively contribute to scientific workflows are capturing market share and redefining industry expectations.
This shift reflects a broader transformation in scientific research, where the value of an ELN is no longer measured by its ability to store data, but by its capacity to generate insights, accelerate decision-making, and integrate seamlessly across the laboratory ecosystem.
Market Transformation and Competitive Realignment
The ELN market in 2026 is characterized by a clear divergence between legacy systems and next-generation platforms.
| Market Dynamic | Legacy ELN Platforms | AI-Driven ELN Platforms |
|---|---|---|
| Core Function | Data storage and documentation | Data interpretation and research intelligence |
| Innovation Pace | Slow | Rapid and continuous |
| Market Position | Declining relevance | Increasing dominance |
| User Value | Administrative support | Strategic scientific enablement |
AI-first platforms are gaining a competitive advantage by embedding intelligence directly into laboratory workflows, enabling researchers to move faster from data generation to actionable insights.
Shifting Global Market Leadership
While North America continues to lead the global ELN market with a 40.50% share in 2026, the balance of power is gradually shifting toward the Asia-Pacific (APAC) region.
| Region | 2026 Market Share | Growth Trajectory | Key Driver |
|---|---|---|---|
| North America | 40.50% | Stable growth | Established R&D ecosystem and high investment levels |
| Asia-Pacific | 20.33% | Rapid expansion | Regulatory mandates and digital transformation |
| Europe | 21.44% | Moderate growth | Compliance-driven modernization |
The APAC region’s projected CAGR of 8.71% reflects the growing influence of regulatory frameworks in countries such as China and India, where pharmaceutical companies are increasingly required to adopt validated digital systems. This regulatory push is accelerating the transition from paper-based processes to fully digital, compliant laboratory environments.
The ELN as Strategic Infrastructure
A critical insight emerging in 2026 is that the ELN is no longer a standalone software solution. Instead, it is becoming a foundational component of laboratory infrastructure—comparable to enterprise resource planning (ERP) systems in business operations.
Modern ELN platforms are expected to deliver a comprehensive set of capabilities that extend far beyond documentation.
| Core Requirement | Strategic Role in 2026 and Beyond |
|---|---|
| Intelligent Assistance | Supports data interpretation, analysis, and reporting |
| System Integration | Unifies ELN, LIMS, and SDMS into a single ecosystem |
| Regulatory Compliance | Ensures adherence to standards such as 21 CFR Part 11 |
| Rapid Deployment | Enables fast implementation and immediate usability |
| User-Centric Design | Promotes adoption through intuitive, familiar interfaces |
These requirements reflect the growing expectation that ELN platforms must function as active participants in the research process rather than passive tools.
The Emergence of the Active Lab Model
The concept of the “Active Lab” represents the next stage in the evolution of scientific research infrastructure. In this model, laboratory systems are interconnected, intelligent, and capable of supporting end-to-end research workflows.
| Laboratory Model | Traditional Approach | Active Lab Model (2030 Vision) |
|---|---|---|
| Data Handling | Fragmented and manual | Unified and automated |
| Decision-Making | Human-driven with delayed insights | AI-assisted and real-time |
| System Integration | Multiple disconnected tools | Fully integrated Lab Operating System |
| Research Speed | Slower due to inefficiencies | Accelerated through automation and insights |
By 2030, the Active Lab model is expected to become the industry standard, fundamentally changing how research organizations operate.
Strategic Imperatives for R&D Leaders
As the market evolves, research and development leaders must adopt a forward-looking approach to ELN selection and implementation.
| Strategic Priority | Implication for Organizations |
|---|---|
| AI Integration | Essential for maintaining competitive advantage |
| Unified Platforms | Reduces inefficiencies and eliminates data silos |
| Compliance Readiness | Critical for regulatory approval and market participation |
| User Adoption | Determines long-term success and return on investment |
| Scalability | Supports future growth and technological evolution |
Organizations that fail to adapt to these priorities risk falling behind in an increasingly data-driven and competitive research environment.
Conclusion: Preparing for the Next Era of Scientific Innovation
The trajectory toward 2030 highlights a fundamental shift in the role of ELN platforms within the scientific ecosystem. No longer confined to documentation, these systems are evolving into intelligent, integrated infrastructures that drive research efficiency, compliance, and innovation.
The transition to AI-enabled Lab Operating Systems will define the next generation of scientific discovery. Organizations that successfully embrace this transformation—by adopting platforms that combine intelligence, integration, and usability—will be best positioned to lead in an era where speed, accuracy, and data-driven insights are paramount.
In this emerging landscape, the ELN is not just a tool but a strategic asset that underpins the future of scientific advancement.
Conclusion
The global Electronic Lab Notebook (ELN) market in 2026 represents far more than a collection of digital tools—it reflects a fundamental transformation in how scientific research is conducted, managed, and scaled. As laboratories across biotechnology, pharmaceuticals, academia, and industrial sectors increasingly transition toward fully digital and data-driven environments, ELNs have evolved into mission-critical infrastructure that underpins innovation, compliance, and operational excellence.
The analysis of the top 10 Electronic Lab Notebook software in the world in 2026 reveals a highly competitive and rapidly evolving ecosystem. Each platform serves a distinct segment of the market, from AI-first innovators such as Genemod, to biologics-focused leaders like Benchling, compliance-driven enterprise solutions such as LabWare and IDBS E-WorkBook, and highly accessible platforms like SciNote and SciSure. This diversity highlights a key reality: there is no one-size-fits-all ELN solution. Instead, the optimal choice depends on an organization’s specific research focus, regulatory requirements, budget constraints, and long-term strategic goals.
The Evolution from Digital Record-Keeping to Intelligent Research Systems
A defining theme across the ELN landscape in 2026 is the transition from passive documentation systems to intelligent, AI-enabled research platforms. Traditional ELNs were primarily designed to replace paper notebooks, offering basic functionality for data capture and storage. However, modern ELNs now play an active role in the scientific process by:
- Interpreting experimental data in real time
- Automating reporting and documentation workflows
- Supporting predictive analytics and hypothesis generation
- Enabling seamless collaboration across global research teams
This shift toward “active” laboratory environments has significantly reduced the gap between data generation and decision-making, allowing organizations to accelerate discovery cycles and improve research outcomes.
Key Factors Defining the Best ELN Software in 2026
Organizations evaluating ELN platforms must consider several critical factors that differentiate leading solutions in today’s market.
| Evaluation Factor | Strategic Importance in 2026 |
|---|---|
| AI and Automation | Enables faster insights, reduced manual work, and smarter workflows |
| Integration Capabilities | Unifies ELN, LIMS, and SDMS to eliminate data silos |
| Regulatory Compliance | Ensures adherence to standards such as 21 CFR Part 11 and Annex 11 |
| Ease of Use | Drives user adoption and long-term productivity |
| Implementation Speed | Impacts “Time to Science” and return on investment |
| Scalability | Supports growth from small labs to global enterprise operations |
Platforms that excel across these dimensions are best positioned to deliver sustained value in increasingly complex research environments.
The Strategic Importance of “Time to Science”
One of the most important insights emerging in 2026 is the concept of “Time to Science”—the time it takes for a laboratory to move from implementation to meaningful scientific output. This metric has become a decisive factor in ELN selection, as organizations seek to minimize downtime and maximize productivity.
Modern cloud-native platforms have dramatically reduced implementation timelines from months to days or weeks, enabling immediate efficiency gains such as:
- Up to 90% reduction in sample search time
- Faster documentation and reporting cycles
- Improved collaboration across distributed teams
In contrast, traditional enterprise systems, while offering deep functionality and compliance capabilities, often require longer deployment periods and higher upfront investment.
Regional and Industry Trends Shaping the ELN Market
The ELN market is also being influenced by significant regional and industry-specific trends.
- North America continues to lead in innovation and market share, driven by strong R&D investment
- Asia-Pacific is emerging as the fastest-growing region, fueled by regulatory mandates and digital transformation
- Europe is advancing through compliance-driven modernization, particularly under Annex 11 and ISO frameworks
At the same time, industry adoption patterns are evolving. While pharmaceutical and biotechnology companies remain dominant, Contract Research Organizations (CROs) are rapidly expanding, creating new demand for collaborative, cloud-based ELN solutions that support real-time data sharing.
The Role of Compliance and Data Integrity
In highly regulated industries, compliance remains a non-negotiable requirement. Leading ELN platforms must support:
- Robust audit trails aligned with the ALCOA+ data integrity model
- Secure electronic signatures and record validation
- Risk-based validation approaches such as Computer Software Assurance (CSA)
- Advanced data provenance capabilities, including blockchain integration in some cases
These features ensure that laboratory data is trustworthy, traceable, and suitable for regulatory inspection, which is essential for organizations operating in pharmaceutical and clinical research environments.
Preparing for the Future: The Rise of the Active Lab
Looking ahead toward 2030, the concept of the “Active Lab” is set to become the industry standard. In this model, ELNs function as intelligent, interconnected systems that:
- Coordinate experimental workflows end-to-end
- Provide real-time insights and recommendations
- Integrate seamlessly with instruments, data systems, and AI models
- Enable fully automated and data-driven research environments
Organizations that successfully adopt these next-generation platforms will gain a significant competitive advantage, as they will be able to innovate faster, reduce operational inefficiencies, and make more informed decisions.
Final Strategic Takeaways for ELN Buyers
For decision-makers evaluating the top Electronic Lab Notebook software in 2026, several key takeaways emerge:
| Strategic Insight | Implication |
|---|---|
| ELN as Infrastructure | Treat ELN as a core component of laboratory operations |
| AI-Driven Capabilities | Prioritize platforms that offer embedded intelligence |
| Unified Ecosystems | Choose solutions that integrate ELN, LIMS, and data management |
| Rapid Deployment | Focus on solutions that deliver quick time-to-value |
| Long-Term Scalability | Ensure the platform can grow with organizational needs |
Closing Perspective
The Electronic Lab Notebook is no longer a simple tool for recording experiments—it is the digital backbone of modern scientific research. As the industry continues to evolve, the platforms that combine intelligence, integration, usability, and compliance will define the future of laboratory innovation.
Selecting the right ELN software in 2026 is therefore not just a technical decision, but a strategic investment in the future of research and development. Organizations that make informed, forward-looking choices today will be the ones leading scientific breakthroughs in the years to come.
If you find this article useful, why not share it with your hiring manager and C-level suite friends and also leave a nice comment below?
We, at the 9cv9 Research Team, strive to bring the latest and most meaningful data, guides, and statistics to your doorstep.
To get access to top-quality guides, click over to 9cv9 Blog.
To hire top talents using our modern AI-powered recruitment agency, find out more at 9cv9 Modern AI-Powered Recruitment Agency.
People Also Ask
What is Electronic Lab Notebook software in 2026?
Electronic Lab Notebook software in 2026 is a digital platform that records, manages, and analyzes lab data, often enhanced with AI, cloud capabilities, and compliance features for modern scientific research.
Why is ELN software important for laboratories?
ELN software improves data accuracy, collaboration, and compliance while reducing manual errors and paper-based inefficiencies in research environments.
What are the top Electronic Lab Notebook software tools in 2026?
Leading ELN tools include Genemod, Benchling, SciNote, Labguru, SciCord, IDBS E-WorkBook, Signals Notebook, Dotmatics, SciSure, and LabWare.
How does AI enhance Electronic Lab Notebook software?
AI helps interpret data, automate reporting, suggest experiments, and accelerate decision-making within laboratory workflows.
What industries use ELN software the most?
Biotechnology, pharmaceuticals, academic research, chemical engineering, and clinical laboratories are the primary users of ELN software.
What is the difference between ELN and LIMS?
ELN focuses on experiment documentation, while LIMS manages workflows, samples, and lab operations. Modern platforms often combine both.
What is a Lab Operating System (LabOS)?
LabOS is an integrated system combining ELN, LIMS, and data management tools into a unified platform for streamlined lab operations.
Which ELN software is best for biotech startups?
Platforms like Genemod, Benchling, and SciSure are ideal for biotech startups due to scalability, AI features, and ease of use.
Which ELN software is best for enterprise laboratories?
LabWare, IDBS E-WorkBook, and Dotmatics are preferred for enterprise labs due to compliance strength and scalability.
What is 21 CFR Part 11 compliance in ELN software?
It is a regulatory standard ensuring electronic records and signatures are secure, traceable, and legally equivalent to paper records.
How much does ELN software cost in 2026?
Pricing varies widely, from free or low-cost plans for small labs to enterprise solutions costing thousands per user annually.
What is the average implementation time for ELN software?
Modern cloud ELNs can be deployed in days or weeks, while enterprise systems may take several months to implement.
What is Time to Science in ELN adoption?
Time to Science refers to how quickly researchers can use the ELN to produce meaningful scientific results after implementation.
Which ELN software offers the fastest deployment?
Cloud-native platforms like Genemod and SciCord offer rapid deployment, often within days or weeks.
What are the benefits of cloud-based ELN software?
Cloud ELNs provide remote access, lower infrastructure costs, scalability, and faster implementation compared to on-premise systems.
Is ELN software suitable for small laboratories?
Yes, many ELNs offer affordable and easy-to-use options tailored for small labs and academic institutions.
What features should you look for in ELN software?
Key features include AI capabilities, compliance support, integration with LIMS, ease of use, scalability, and data security.
How does ELN software improve data integrity?
ELNs ensure data integrity through audit trails, secure access controls, and adherence to standards like ALCOA+.
What is ALCOA+ in laboratory data management?
ALCOA+ defines data integrity principles such as being attributable, legible, contemporaneous, original, accurate, complete, consistent, enduring, and available.
Can ELN software integrate with lab instruments?
Yes, many ELNs integrate with lab instruments to capture data automatically and reduce manual input errors.
Which ELN software is best for chemistry research?
Signals Notebook and Dotmatics are strong choices for chemistry-focused labs due to specialized tools and visualization features.
What are the limitations of ELN software?
Some ELNs may have high costs, steep learning curves, limited customization, or lack advanced AI capabilities.
How secure is ELN software in 2026?
Modern ELNs use encryption, role-based access, and compliance certifications like ISO 27001 and SOC 2 to ensure data security.
What is the role of AI in drug discovery within ELNs?
AI helps identify targets, optimize compounds, and generate new molecules, accelerating drug discovery processes.
What is Shadow AI in laboratory environments?
Shadow AI refers to the use of external AI tools outside official systems, which is now being replaced by integrated ELN AI features.
Which region leads the ELN market in 2026?
North America leads the market, while Asia-Pacific is the fastest-growing region due to regulatory and digital transformation.
How does ELN software support regulatory compliance?
ELNs provide audit trails, electronic signatures, and validated workflows to meet regulatory standards.
What is the ROI of implementing ELN software?
ROI includes improved efficiency, faster research cycles, reduced errors, and better data management.
Can ELN software replace paper lab notebooks completely?
Yes, modern ELNs fully replace paper notebooks while offering additional features like searchability and collaboration.
What is the future of Electronic Lab Notebook software?
The future lies in AI-driven, integrated platforms that enable fully automated, data-driven “Active Lab” environments.
Sources
Mordor Intelligence Lab Manager LabOS Scispot SciCord IntuitionLabs Towards Healthcare Fact.MR GlobeNewswire SNS Insider Fortune Business Insights Technology Networks News Medical Benchling Genemod G2 Gitnux FitGap SciNote DevOpsSchool Research.com SourceForge IDBS Dotmatics eCFR QT9 Software SimplerQMS LabArchives Intellivon































![Writing A Good CV [6 Tips To Improve Your CV] 6 Tips To Improve Your CV](https://blog.9cv9.com/wp-content/uploads/2020/06/2020-06-02-2-100x70.png)


